Abstract
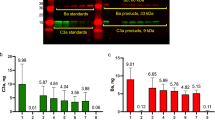
The ability of H to inhibit formation of the alternative pathway C3 convertase differs on the non-activating cells sheep erythrocytes (Es) and on the activating cells rabbit erythrocytes (Er) or desialated sheep erythrocytes (Edes). C3 convertase sites are converted to C5 convertase sites by deposition of additional C3b molecules and C5 binds to cell-bound C3b in a reaction that is inhibited by H. C5 convertase sites P(C3b)nBb were formed on Es, Er, Edes and revealed by incubation of the cells with purified C5 and with rat serum treated with KSCN and hydrazine. When incremental amounts of H were added to the reaction, a dose-dependent inhibition of C5 utilization was observed. The amount of H that inhibited 50% of C5 utilization was the same for a given cell regardless of the number of C5 convertase sites that were expressed on the cell. However the 50% inhibitory dose of H was 92 ng/107 EsP(C3b)nBb, 87 ng/107 EdesP(C3b)nBb while it was at least 380 ng/107 ErP(C3b)nBb. These results suggest that H competes with C5 for uptake within the C5 convertase site and that the regulatory effect of H on C5 cleavage depends on the surface to which the convertase is bound.
Similar content being viewed by others
References
M.D. Kazatchkine andU.E. Nydegger,The human alternative complement pathway: biology and immunopathology of activation and regulation, Prog. Allergy.30, 193–234 (1982).
M.R. Daha, D.T. Fearon andK.F. Austen,C3 requirements for formation of alternative pathway C5 convertase, J. Immun.117, 630–634 (1976).
W. Vogt, G. Schmidt, B. Von Buttlar andL. Dieminger,A new function of the activated third component of complement: binding to C5, an essential step for C5 activation, Immunology34, 29–40 (1978).
D.E. Isenman, E.R. Podack andN.R. Cooper,The interaction of C5 with C3b in free solution: a sufficient condition for cleavage by a fluid phase C3/C5 convertase, J. Immun.124, 326–331 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fischer, E., Jouvin, MH. & Kazatchkine, M. The ability of H to modulate C5 utilization by the cell-bound alternative pathway C5 convertase differs on sheep and rabbit erythrocytes. Agents and Actions 13, 430–431 (1983). https://doi.org/10.1007/BF02176406
Issue Date:
DOI: https://doi.org/10.1007/BF02176406