Abstract
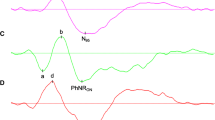
Pattern electroretinograms (P-ERGs) and pattern visual evoked potentials (P-VEPs) were recorded at three luminance levels and five different check sizes in a group of 16 control subjects before and after the oral administration of levodopa. At the lower luminance levels, significant decrease in P-ERG and P-VEP latencies were found. For P-VEPs the latency changes occurred only at small check sizes. No changes were observed in control experiments without levodopa administration. Our results show that levodopa-induced changes even occur at the retinal level and support a dopaminergic involvement in light and dark adaptation. Our observations are in agreement with a VEP delay found in Parkinson's disease and with a VEP latency increase in rats after dopamine depletion.
Similar content being viewed by others
References
Bodis-Wollner I, Yahr MD (1978) Measurements of visual evoked potentials in Parkinson's disease. Brain 101:661–671
Bodis-Wollner I, Yahr MD, Mylin L, Thorton J (1981) Dopaminergic deficiency and delayed visual evoked potentials in humans. Ann Neurol 11:478–483
Borbe HO, Fehske JK, Müller WE, Nover A, Wollert U (1982) The demonstration of several neurotransmitter and drug receptors in human retina. Comp Biochem Physiol 72:117–119
Brainard GC, Morgan WW (1987) Light-induced stimulation of retinal dopamine: a dose-response relationship. Brain Res 424:199–203
Calne DB, Reid JL (1982) Antiparkinson drugs, pharmacological and antitherapeutical aspects. Drugs 4:49–74
Cleland BG, Levick WR (1974) Brisk and sluggish concentrically organized ganglion cells in the cat's retina. J Physiol 240:421–456
Cowey A, Rolls ET (1974) Human cortical magnification factor and its relation to visual acuity. Exp Brain Res 21:447–454
Dawis SM, Niemeyer G (1988) Similarity and diversity of monoamines in their effects on the standing potential, light peak and electroretinogram of the perfused cat eye. Clin Vis Sci 3:109–118
Dinner DS, Lüders H, Hanson M, Lesser RP, Klem G (1985) Pattern evoked potentials (PEPs) in Parkinson's disease. Neurology (NY) 35:610–613
Dyer RS, Howell WE, MacPhail RC (1981) Dopamine depletion slows retinal transmission. Exp Neurol 71:326–340
Ehle AL, Stewart M, Lellelid NE, Leventhal NA (1982) Normal checkerboard pattern reversal evoked potentials in parkinsonism. Electroencephalogr Clin Neurophysiol 54: 336–338
Ellis CJK, Allen GJ, Marsden CD, Ikeda H (1987) Electroretinographic abnormalities in idiopathic Parkinson's disease and the effect of levodopa administration. Clin Vis Sci 1:347–355
Filip V, Balik J (1978) Possible indication of dopaminergic blockade in man by electroretinography. Int Pharmacopsychiatry 13:151–156
Frederick JM, Rayborn ME, Laties A, Lam DMK, Hollyfield JD (1982) Dopaminergic neurons in the human retina. J Comp Neurol 210:65–79
Gawel MJ, Das P, Vincent S, Rose C (1982) Visual and auditory evoked responses in patients with Parkinson's disease. J Neurol Neurosurg Psychiatry 44:227–232
Gottlob I, Schneider E, Heider W, Skrandies W (1987) Alteration of visual evoked potentials and electroretinograms in Parkinson's disease. Electroencephalogr Clin Neurophysiol 66:349–357
Hansch EC, Syndulko K, Cohen SN, Goldberg ZI, Potvin AR, Tourtellote WW (1982) Cognition in Parkinson's disease: an event-related potential perspective. Ann Neurol 11:599–607
Iuvone PM, Galli CL, Garrison-Gund CK, Neff NH (1978) Light stimulated tyrosine hydroxylase activity and dopamine synthesis in retinal amacrine neurons. Science 202: 901–902
Jaffe MJ, Bruno G, Campell G, Lavine RA, Karson CN, Weinberger DR (1987) Ganzfeld electroretinographic findings in parkinsonism: untreated patients and the effect of levodopa intravenous infusion. J Neurol Neurosurg Psychiatry 50:847–852
Jagadeesh JM, Sanchez R (1981) Effect of apomorphine on the rabbit electroretinogram. Invest Ophthalmol Vis Sci 21:620–625
Jagadeesh JM, Lee HC, Salazar-Bookaman M (1980) Influence of chlorpromazine on the rabbit electroretinogram. Invest Ophthalmol Vis Sci 19:1449–1456
Jensen RJ, Daw NW (1984) Effect of dopamine antagonists on receptive fields of brisk cells and directionally selective cells in the rabbit retina. J Neurosci 4:2972–2985
Maguire GW, Smith EL (1985) Cat retinal ganglion cell receptive-field alterations after 6-hydroxydopamine induced dopaminergic amacrine cell lesions. J Neurophysiol 53:1431–1443
Marx M, Bodis-Wollner I, Bobak P, Harnois C, Mylin L, Yahr M (1986) Temporal frequency-dependent VEP changes in Parkinson's disease. Vision Res 26:185–193
Odom JV, Maida TM, Dawson WW (1983) Pattern evoked retinal response (PERR) in humans: effects of spatial frequency, temporal frequency, luminance and defocus. Curr Eye Res 2:99–108
Onofrj M, Bodis-Wollner I, Ghilardi MF, Marx M, Glover A (1986) Pattern vision in monkeys with parkinsonism: a stimulus specific effect of MPTP on retinal and cortical responses. In: Markey SP, Castagnoli N Jr, Trevor AJ, Kopin IJ (eds) MPTP — a neurotoxin producing a parkinsonian syndrome. Academic Press, New York, pp 683–688
Parkinson D, Rando RR (1983) Effects of light on dopamine metabolism in the chick retina. J Neurochem 40:39–47
Parkinson D, Rando RR (1983) Effect of light on dopamine turnover and metabolism in rabbit retina. Invest Ophthalmol Vis Sci 24:384–388
Proll MA, Morgan WW (1982) Adaptation of retinal dopamine neuron activity in light-adapted rats to darkness. Brain Res 241:359–361
Schneider T, Zrenner E (1987) The effect of fluphenazine on rod-mediated retinal responses. Doc Ophthalmol 65:287–296
Strachil M, Perwein J (1969) The inhibition of retinal ganglion cells by catecholamines and γ-aminobutyric acid. Pflüger's Arch 321:45–54
Tartaglione A, Pizio N, Bino G, Spadavecchia L, Favale E (1984) VEP changes in Parkinson's disease are stimulus dependent. J Neurol Neurosurg Psychiatry 47: 305–307
Thier P, Alder V (1983) Action of iontophoretically applied dopamine on cat retinal ganglion cells. Brain Res 292:109–121
Thierry AM, Stinus L, Blanc G, Glowinski J (1973) Some evidence for the existence of dopaminergic neurons in the rat cortex. Brain Res 50:230–234
Yaar I (1980) The effect of levodopa treatment on the visual evoked potentials in parkinsonian patients. Electroencephalogr Clin Neurophysiol 50:267–274
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gottlob, I., Weghaupt, H., Vass, C. et al. Effect of levodopa on the human pattern electroretinogram and pattern visual evoked potentials. Graefe's Arch Clin Exp Ophthalmol 227, 421–427 (1989). https://doi.org/10.1007/BF02172892
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02172892