Abstract
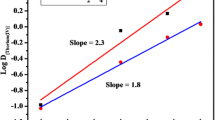
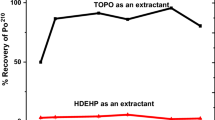
A method based on solvent extraction has been developed for the radiochemical separation of Zn/II/ from other elements employing 1,2,3-benzotriazole as a complexing agent and n-heptanol as an extractant. Optimum conditions for the extraction have been established. The effect of various anions and cations on the extraction coefficient value of Zn/II/ has been studied. The stoichiometry of the metal to reagent determined by the method of substoichiometric extraction and slope-ratio method was found to be 1∶2.
Similar content being viewed by others
References
S. Subramanian, Z.R. Turel,J. Radioanal. Nucl. Chem., Lett., 103 /1986/ 365.
I.A. Mendes, Z.R. Turel,J. Radioanal. Nucl. Chem., Lett., 96 /1985/ 343.
S. Subramanian, Z.R. Turel,J. Radioanal. Nucl. Chem. Lett., 105 /1986/ 117.
I.A. Mendes, Z.R. Turel, J. Radioanal. Nucl. Chem., Lett., 104 /1986/ 43.
I.A. Mendes, Z.R. Turel,J. Radioanal. Nucl. Chem., Lett., 105 /1986/ 201.
A.I. Vogel, Textbook of Quantitative Inorganic Analysis, Longmans Green and Co. Ltd., London, 1975.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kapadia, J., Turel, Z.R. Solvent extraction of Zn/II/ with 1,2,3-benzotriazole into n-hepatanol. Journal of Radioanalytical and Nuclear Chemistry Letters 118, 15–22 (1987). https://doi.org/10.1007/BF02165650
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02165650