Abstract
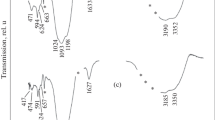
Thermolysis of cesium bis (citrato) ferrate (III) trihydrate, Cs3[Fe(C6H5O7)2].3H2O, has been studied in the temperature range of 373–1073 K in static air atmosphere, using Mössbauer and infrared spectroscopy and thermogravimetric methods. The anhydrous complex decomposed into an iron (II) intermediate at 453 K and α-Fe2O3 of varying particle sizes in successive stages. Finally a solid state reaction between α-Fe2O3 and cesium carbonate/oxide gives fine particles of cesium ferrite (CsFeO2) at a temperature lower than that of conventional ceramic method.
Similar content being viewed by others
References
A. Srivastva, P. Singh, V.G. Gunjikar, A.P.B. Sinha,Thermochim. Acta, 86 (1985) 77.
B.S. Randhawa,J. Radioanal. Nucl. Chem., Lett., 201 (1995) 57.
A.S. Brar, B.S. Randhawa,J. Phys. (Paris), 44 (1983) 1345.
B.S. Randhawa, Sandeep Kaur, P.S. Bassi,Indian J. Chem., 28A (1989) 46.
K. Nakamoto, Infrared Spectra of Inorganic and Coordination Compounds, 2nd edn., J. Wiley Interscience, New York, 1970.
A. Vértes, L. Korecz, K. Burger, Mössbauer Spectroscopy, Elsevier, New York, 1979, p. 47.
V.G. Silnichenko,Chem. Abs., 61 (1964) 3899 h.
O. Liesche,Z. Phys. Chem., 94 (1920) 663,Chem. Abst., 14 (1920) 2442.
P.K. Gallagher, C.R. Kurkjian,Inorg. Chem., 5 (1966) 214.
W. Miesel, G. Kreysa,Z. Anorg. Allg. Chem., 395 (1972) 31.
I.F. Alenchikova, B.E. Dzevitskii, V.S. Neporezov, N.N. Savvateev, V.F. Sukhoverkhov,Russian J. Inorg. Chem., 20 (1975) 1198.
R.A. Nyquist, R.O. Kagel, Infrared Spectra of Inorganic Compounds, Academic Press, New York, 1971.
Langes Handbook of Chemistry (Ed. by J.A. Dean), 11th Edn., McGraw-Hill Book Co., New York, 1973.
H. Irving, R.J.P. Williams, D.J. Ferrett, A.E. Williams,J. Chem. Soc., (1954) 3494.
L. Erdey, S. Gal, G. Liptay,Talanta, 11 (1964) 913.
T. Ichida, T. Shinjo, Y. Bando, T. Takada,J. Phys. Soc. Japan, 29 (1970) 795, 1109.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Randhawa, B.S. Mössbauer study of the thermolysis of cesium bis (citrato) ferrate (III) trihydrate. Journal of Radioanalytical and Nuclear Chemistry Letters 212, 43–50 (1996). https://doi.org/10.1007/BF02165450
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02165450