Abstract
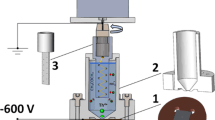
Preparation of thin film deposits of lanthanide, thorium and uranium oxides has been studied by chemical vapor deposition (CVD) method using β-diketonate metal chelates with 2,2,6,6-tetramethyl-3,5-heptanedione and some reactant gases as starting materials. The deposition process was carried out using a special apparatus designed for the CVD method at atmospheric pressure and temperatures as low as 400–600°C.As a result, it was demonstrated that each chelate used was well suited for the above purpose by its high volatility and reactivity with the reactant, especially with water vapor.
Similar content being viewed by others
References
K. F. JENSEN, Microelectronics Processing, D. W. HESS, K. F. JENSEN (Eds), American Chemical Society, Washington 1989, p. 199.
K. J. EISENTRAUT, R. E. SIEVERS, J. Am. Chem. Soc., 87 (1965) 5254.
J. E. SICRE, J. T. DUBOIS, K. J. EISENTRAUT, R. E. SIEVERS, J. Amer. Chem. Soc., 91 (1969) 3476.
R. E. SIEVERS, E. SADLOWSKI, Science, 201 (1978) 217.
M. SAKANOUE, R. AMANO, Transplutonium 1975, W. MULLER, R. LINDNER (Eds), North-Holland Publishing Company, Amsterdam 1976, p. 123.
H. YAMANE, H. KUROSAWA, T. HIRAI, Chem. Lett., (1988) 939.
A. D. BERRY, D. K. GASKILL, R. T. HOLM, E. J. CUKAUSKAS, R. KAPLAN, R. L. HENRY, Appl. Phys. Lett., 52 (1988) 1743.
K. NATIO, N. KAMEGASHIRA, Adv. Nucl. Technol. 9 (1976) 99.
H. R. HOEKSTRA, S. SIEGEL, F. X. GALLAHHER, J. Inorg. Nucl. Chem., 32 (1970) 3237.
F. WIEGEL, H. R. HOEKSTRA, The Chemistry of the Actinide Elements, J. J. KATZ, G. T. SEABORG, L. R. MORSS (Eds), Chapman and Hall, New York 1986, p. 256.
J. C. SPIRLET, O. VOGT, Handbook on the Physics and Chemistry of the Actinides Vol. 1, A. J. FREEMAN, G. H. LANDER (Eds), Elsevier Science Publishers B. V., Amsterdam 1984, p. 79.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shiokawa, Y., Amano, R., Nomura, A. et al. Preparation of lanthanide, thorium and uranium oxide films by chemical vapor deposition using β-diketone chelates. Journal of Radioanalytical and Nuclear Chemistry, Articles 152, 373–380 (1991). https://doi.org/10.1007/BF02104690
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02104690