Abstract
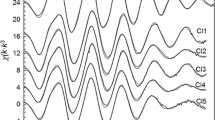
Mössbauer data as well as infrared and magnetic susceptibility results indicate that Fe(III)-phthalates are high spin ferric complexes with a combination of trinuclear oxo-bridged and monomer structures. Values obtained for the isomer shift and quadrupole splitting parameters suggest that different counter ions, such as OH, NO3, and Na, leave the Fe(III) ions in two very distorted octahedral arrangements, although they scarcely affect the electron densities at the iron nuclei.
Similar content being viewed by others
References
B.O. West, Polyhedron 8 (1989) 219.
Ch. T. Dziobkowski, J.T. Wrobleski, D.B. Brown, Inorg. Chem. 20 (1981) 671.
V. Vancina, T.A. Himdan, H. Bilinski, M. Miljak, D. Hanzel and Darko Hanzel, to be published.
S.M. Gorun, G.C. Papaefthyiou, R.B. Frankel, S.J. Lippard, J. Am. Chem. Soc. 109 (1987) 4244.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hanzel, D., Hanzel, D., Bilinski, H. et al. A study of trinuclear iron(III) O-phthalates. Hyperfine Interact 53, 339–343 (1990). https://doi.org/10.1007/BF02101063
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02101063