Abstract
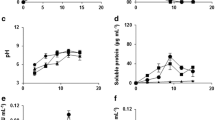
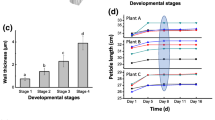
Cell-free extracts and culture fluids ofSchizophyllum commune were assayed for enzymatic activity effecting the degradation of an alkali-insoluble cell-wall component of this mushroom, a glucan containingβ-(1→3) linkages (R-glucan). The activity of R-glucanase as determinedin vitro with isolated R-glucan as a substrate was found to increase from the onset of pileus formation, a process accompanied by R-glucan degradation in the mycelium. This R-glucanase activity is influenced by the presence of glucose in the culture medium, probably through a mechanism by which glucose represses synthesis of the enzyme.
A morphological mutant (cup mutant) producing no pilei and exhibiting a lower degradation of R-glucanin vivo, produced levels of R-glucanase comparable to those of the wild-type stock and gave even higher levels in young cultures. The difference between the wild-type stock and the cup mutant with respect to degradation of R-glucan during development is most probably to be sought in the structure of the cell wall, the R-glucan in isolated cell walls of the cup mutant being less susceptible to enzymatic attack. High resistance to R-glucanase activity was also encountered in certain cell-wall preparations of the wild-type stock e.g. in those prepared from developing pilei. This suggests that cell-wall glucan degradation during pileus formation is controlled by both the level of R-glucanase, as influenced by glucose in the medium, and differences in protection of R-glucan in the cell wall against enzymatic attack.
Similar content being viewed by others
References
Brock, T. D. 1965.β-Glucanase of yeast. Biochem. Biophys. Res. Comm.19 623–629.
Chesters, C. G. C. andBull, A. T. 1963a. The enzymic degradation of laminarin. 1. The distribution of laminarase among micro-organisms. Biochem. J.86 28–31.
Chesters, C. G. C. andBull, A. T. 1963b. The enzymic degradation of laminarin. 2. The multicomponent nature of fungal laminarases. Biochem. J.86 31–38.
Clarke, A. E. andStone, B. A. 1965.β-Glucan hydrolases fromAspergillus niger. Isolation of aβ-(1→4)-glucan hydrolase and some properties of theβ-(1→3)-glucan hydrolase components. Biochem. J.96 793–801.
Fairbairn, N. J. 1953. A modified anthrone reagent. Chem. Ind. (London)1953: 86.
Hauge, J. G., MacQuillan, A. M., Cline, A. L. andHalvorson, H. O. 1961. The effect of glucose repression on the level of ribosomal-boundβ-glucosidase. Biochem. Biophys. Res. Comm.5 267–269.
Jermyn, M. A. 1965. Fungal cellulases. XI. The nature of the inductive process for arylβ-glucosidase inStachybotrys atra. Australian J. Biol. Sci.18 387–415.
Johnston, I. R. 1965. The composition of the cell wall ofAspergillus niger. Biochem. J.96 651–658.
Kreger, D. R. 1954. Observations on cell walls of yeasts and some other fungi by X-ray diffraction and solubility tests. Biochim. Biophys. Acta13 1–9.
Lowry, O. H., Rosebrough, N. J., Farr, A. L. andRandall, R. J. 1951. Protein measurement with the Folin reagent. J. Biol. Chem.193 265–275.
Reese, E. T. andMandels, M. 1959.β-d-1,3 Glucanases in fungi. Can. J. Microbiol.5 173–185.
Reese, E. T. andMandels, M. 1963. Enzymatic hydrolysis ofβ-glucans. p. 197–234.In E. T. Reese, [ed.], Advances in enzymic hydrolysis of cellulose and related materials. Pergamon Press, New York.
Russell, D. W., Sturgeon, R. J. andWard, V. 1964. Preparation and properties of a hyphal wall fraction fromPithomyces chartarum. J. Gen. Microbiol.36 289–296.
Skujins, J. J., Potgieter, H. J. andAlexander, M. 1965. Dissolution of fungal cell walls by a Streptomycete chitinase andβ-(1→3) glucanase. Arch. Biochem. Biophys.111 358–364.
Wessels, J. G. H. 1965. Morphogenesis and biochemical processes inSchizophyllum commune Fr. Wentia13 1–113.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wessels, J.G.H. Control of cell-wall glucan degradation during development inSchizophyllum commune . Antonie van Leeuwenhoek 32, 341–355 (1966). https://doi.org/10.1007/BF02097484
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02097484