Abstract
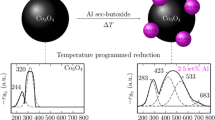
Decomposition of cobalt nitrate on alumina in air or hydrogen leads to the formation of different cobalt oxide phases. Chemisorption of H2 and IR spectra of adsorbed CO show that for the catalyst pretreated in H2 the reduction to metallic cobalt occurs at T>573 K and results in lower metal dispersity. Formation of cobalt particles of different surface structures is proposed depending on the pretreatment mode.
Similar content being viewed by others
References
P. Arnoldy, J. A. Moulijn:J. Catal.,93, 38 (1985).
A. Lapidus, A. Krylova, V. Kazanskii, V. Borovkov, A. Zaitsev, J. Ratousky, A. Zukal, M. Jancalkova:Appl. Catal.,73, 65 (1991).
R. L. Chin, D. Hercules:J. Phys. Chem.,86, 360 (1982).
M. A. Stranick, M. Houalla, D. M. Hercules:J. Catal.,103, 151 (1987).
T. S. Dorokhina, A. V. Zaitsev, V. Yu. Borovkov, V. B. Kazanskii:Kinet. Katal.,32, 1499 (1992).
A. Palazov, G. Kadinov, Ch. Bonev, D. Shopov:Surf. Sci.,188, 505 (1987).
N. Sheppard, T. T. Nguyen:Advances in Infrared and Raman Spectroscopy, Vol. 5, Chap. 2, Eds. R. J. H. Clark and R. E. Hester. Heyden, London 1978.
J. Ansorge, H. Foerster:Z. Phys. Chem. N. F.,118, 113 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Todorova, S., Zhelyazkov, V. & Kadinov, G. IR, TPR and chemisorption study of alumina-supported cobalt catalysts. React Kinet Catal Lett 57, 105–110 (1996). https://doi.org/10.1007/BF02076127
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02076127