Abstract
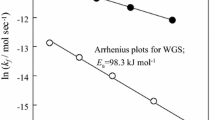
The water-gas shift reaction was studied at 563–638 K over an industrial ferrochrome catalyst. The reaction rate data could be described by first order kinetics coupled to a diffusional resistance model for isothermal pellets. The model parameters were determined by simultaneous nonlinear regression.
Abstract
Реакцию водяного газа исследовали на промышленном феррохромном катализаторе при 563–683 К. Данные скорости реакции подчиняутся кинетике первого порядка совместно с моделью диффузионного сопротивления для изотермических гранул. Параметры модели были определены с помошью нелинейных регрессий.
Similar content being viewed by others
References
T. Salmi, L.-E. Lindfors, S. Boström: Chem. Engng. Sci.,41, 929 (1986).
J. M. Moe: Chem. Engng. Prog.,58, 33 (1962).
R. C. Reid, J. M. Prausnitz, T. K. Sherwood: The Properties of Gases and Liquids, 3rd Ed. McGraw-Hill New York 1977.
E. L. Furen, D. V. Gernet, T. A. Semenova, M. P. Shmigirovskaya: Kinet. Katal.,12, 1283 (1971).
C. Bokhoven, H. Hoogschagen: J. Chem. Phys.,21, 159 (1953).
G. C. Chinchen, R. H. Logan, M. S. Spencer: Appl. Catal.,12, 69 (1984).
D. M. Ruthven: Can. J. Chem. Engng.,47, 327 (1969).
S. Vajda, P. Valko: Reproche, Regression Programs for Chemical Engineers Manual, Budapest 1985.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Salmi, T. Simultaneous determination of kinetic and diffusion parameters for the water-gas shift reaction. React Kinet Catal Lett 41, 1–5 (1990). https://doi.org/10.1007/BF02075473
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02075473