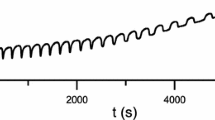
Abstract
The heat output (Q) during the “induction period” of a Belousov-Zhabotinskii oscillating reaction usually evolves at a constant rate /6/; we observe in the BrO −3 /Ce4+/Cyclohexanone system a sudden change in the slope of the function Q=f(t), which corresponds to the beginning of the “quasistationary phase” /3/.
Abstract
Выход тепла (Q) во время “индукционного периода” осциллирующей реакции Белоусова-жаботинского обычно происходит при постоянной скорости; в системе BrO −3 /Ce+4/цикдогексанон происходит неожиданное изменеие наклона функции Q=f(t), что соотвествует началу “квазистационарной фазы”.
Similar content being viewed by others
References
V.J. Farage, Ph. Sroot, D. Janjic: Helv. Chim. Acta,60, 231 (1977).
V.J. Farage, D. Janjic: Helv. Chim. Acta,61, 1539 (1978).
V.J. Farage, D. Janjic: Helv. Chim. Acta,63, 433 (1980).
E. Kőrös, M. Orbán, Zs. Nagy: Nature, Phys. Science,242, 30 (1973).
E. Kőrös, M. Orbán, Zs. Nagy: J. Phys. Chem.,77, 3122 (1973).
E. Kőrös, M. Orbán, Zs. Nagy: Therm. Anal. Proc. Int. Conf.,1, 131 (1974).
E. Kőrös, M. Orbán, Zs. Nagy: Acta Chim. (Budapest),100, 449 (1979).
H.G. Busse: Nature, Phys. Science,233, 137 (1971).
G. Schmitz: Can. J. Chem.,55, 3147 (1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Farage, V.J., Janjic, D. Calorimetric study of the BrO −3 /Ce4+/cyclohexanone and BrO −3 /Ce4+/cyclopentanone oscillating systems. React Kinet Catal Lett 15, 487–491 (1981). https://doi.org/10.1007/BF02074154
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02074154