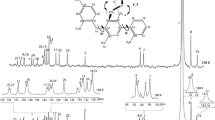
Abstract
The kinetics of formation and cleavage of hydrogen bonds in complexes of carboxylic acids with proton acceptors have been studied by PMR spectroscopy over the range −100 to 150°C. The activation parameters of the processes have been measured.
Abstract
По спектрам ЯМР1H в интервале −100° ÷–155°C исследована кинетика процессов образования и разрыва водородной связи в комплексах карбоновых кислот с акцепторами протона. Пзмерены параметры активации зтих процессов.
Similar content being viewed by others
References
M. Eigen: Chem. Eng. News,38 (1963).
F. Bader, R. Plass: Ber. Bunsenges. Phys. Chem.,75, 353 (1971).
E. Grunwald, E. R. Ralph: Acc. Chem. Res.,4, 107 (1971).
A. Fratiello, R. E. Schster, G. A. Vidulich, J. Bragin, D. Lin: J. Amer. Chem. Soc.,95, 639 (1973).
M. T. Rogers, J. C. Woodbrey: J. Phys. Chem.,66, 540 (1962).
A. A. Mashkovskii, V. P. Glazunov, S. E. Odinokov: Zh. Prikl. Spektrosk.,20, 852 (1974).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Golubev, N.S., Denisov, G.S. PMR study of the lifetime of complexes with a strong hydrogen bond at low temperatures. React Kinet Catal Lett 4, 87–92 (1976). https://doi.org/10.1007/BF02073975
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02073975