Summary
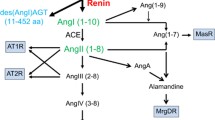
1. Angiotensin II is a well-known vasopressive octapeptide that is the principal end-product of the renin-angiotensin system. In addition to its tonic effect on vascular smooth muscle cells, it also stimulates aldosterone secretion from the adrenals and promotes sodium reabsorption through renal tubular cells.
2. These physiological functions have been appreciated for some time, but as details of the molecular and cell biology of the angiotensin response mechanism become understood, it is increasingly apparent that the hormone has a much broader repertoire. Its functional variability is made possible by (i) different enzymatic routes for its generation, (ii) different receptors distributed in different tissues, (iii) different mechanisms for receptor regulation, and (iv) different signal transduction pathways.
3. This insight is the direct consequence of advances in pharmacology that led first to inhibitors of angiotensin converting enzyme and later to angiotensin II receptor antagonists. This review looks at the current status of angiotensin biochemistry and physiology and provides a basis for anticipation of future developments.
Similar content being viewed by others
References
Alexander, D. R. (1990). The role of phosphatases in signal transduction.New Biologist 21049–1062.
Bernstein, K. E., and Berk, B. C. (1993). The biology of angiotensin II receptors.Am. J. Kidney Dis. 22745–754.
Bhat, G. J., Thekkumkara, T. J., Thomas, W. G., Conrad, K. M., and Baker, K. M. (1994). Angiotensin II stimulatessis-inducing factor-like DNA binding activity.J. Biol. Chem. 26931443–31449.
Bihoreau, C., Monnot, C., Davies, E., Teutsch, B., Bernstein, K. E., Corvol, P., and Clauser, E., (1993). Asp74 mutants of the rat vascular angiotensin II receptor (AT1) display abnormal binding and transmembrane signalling.Proc. Natl. Acad. Sci. USA 905133–5137.
Bumpus, F. M., Schwartz, H., and Page, I. H. (1957). Synthesis and pharmacology of the octapeptide angiotensin.Science 125886–887.
Bünning, P., and Riordan, J. F. (1983). Activation of angiotensin converting enzyme by monovalent anions.Biochemistry 22110–116.
Byers, L. D., and Wolfenden, R. (1973). Binding of the two-product analog benzylsuccinic acid by carboxypeptidase A.Biochemistry 122070–2078.
Campbell, P. T., Hnatowich, M., O'Dowd, B. F., Caron, M. G., Lefkowitz, R. J., and Hausdorff, W. P. (1991). Mutations of the human β2-adrenergic receptor that impair coupling to Gs interfere with receptor down-regulation but not sequestration.Mol. Pharmacol. 39192–198.
Catt, K. J., Hunyady, L., and Balla, T. (1991). Second messengers derived from inositol lipids.J. Bioenerg. Biomemb. 237–27.
Clauser, E., Gaillard, I., Wei, L., and Corvol, P. (1989). Regulation of angiotensinogen gene.Am. J. Hypertens. 2403–410.
Cushman, D. W., Cheung, H. S., Sabo, E. F., and Ondetti, M. A. (1977). Design of potent competitive inhibitors of angiotensin-converting enzyme. Carboxyalkanoyl and mercaptoalkanoyl amino acids.Biochemistry 165483–5491.
Dasso, L. L. T., and Taylor, C. W. (1994). Interactions between Ca2+-mobilizing receptors and their G proteins in hepatocytes.J. Biol. Chem. 2698647–8652.
Doolittle, R. F. (1983). Angiotensinogen is related to the antitrypsin-antithrombin—ovalbumin family.Science 222417–419.
Dzau, V. J. (1987). Possible prorenin activating mechanism in the blood vessel wall.J. Hypertens. 5(Suppl.2):S15-S18.
Dzau, V. J. (1994). Cell biology and genetics of angiotensin in cardiovascular disease.J. Hypertens. 12(Suppl.4):S3-S10.
Dzau, V. J., Mukoyama, M., and Platt, R. E. (1994). Molecular biology of angiotensin receptors: target for drug research?J. Hypertens. 12(Suppl.2):S1-S5.
Dzau, V. J., Sasamura, H., and Hein, L. (1993). Heterogeneity of angiotensin synthetic pathways and receptor subtypes: Physiological and pharmacological implications.J. Hypertens.11(Suppl.3):S13-S18.
Ehlers, M. R. W., and Riordan, J. F. (1990). Angiotensin-converting enzyme. Biochemistry and molecular biology. In Laragh, J. H., and Brenner, B. M. (eds.),Hypertension: Pathophysiology, Diagnosis, and Management, Raven Press, New York, pp. 1217–1231.
Ehlers, M. R. W., and Riordan, J. F. (1991). Angiotensin-converting enzyme: Zinc- and inhibitor-binding stoichiometries of the somatic and testis isozymes.Biochemistry 307118–7126.
Furakawa, Y., Kishimoto, S., and Nishikawa, K. (1982). Hypotensive imidazole derivatives and Hypotensive imidazole-5-acetic acid derivatives. Patents issued to Takeda Chemical Industries Ltd. July 20, 1982, and October 19, 1982 respectively, U.S. Patents 4,340,598 and 4,355,040, Osaka, Japan.
Griendling, K. K., and Alexander, R. W. (1993). The angiotensin (AT1) receptor.Semin. Nephrol. 13558–566.
Griendling, K. K., Tsuda, T., Berk, B. C., and Alexander, R. W. (1989). Angiotensin II stimulation of vascular smooth muscle cells.Am. J. Hypertens. 2659–665.
Howard, T. E., Shai, S.-Y., Langford, K. G., Martin, B. M., and Bernstein, K. E. (1990). Transcription of testicular angiotensin-converting enzyme (ACE) is initiated within the 12th intron of the somatic ACE gene.Mol. Cell. Biol. 104294–4302.
Huckle, W. R., and Earp, H. S. (1994). Regulation of cell proliferation and growth by angiotensin II.Prog. Growth Factor Res. 5177–194.
Hunyady, L., Baukal, A. J., Balla, T., and Catt, K. J. (1994). Independence of type I angiotensin II receptor endocytosis from G protein coupling and signal transduction.J. Biol. Chem. 26924978–24804.
Hunyady, L., Bor, M., Balla, J., and Catt, K. J. (1994). Identification of a cytoplasmic Ser-Thr-Leu motif that determines agonist-induced internalization of the AT1 angiotensin receptor.J. Biol. Chem. 26931378–31382.
Inagami, T. (1993). Renin: Purification, structure and function. In Robertson, J. I. S., and Nicholls, M. G. (eds.),The Renin Angiotensin System, Gower Medical Publishing, London, pp. 4.1–4.17.
Inagami, T. (1994). The renin-angiotensin system.Essays Biochem. 28147–164.
Jard, S., Cantau, B., and Jakobs, K. H. (1981). Angiotensin II andα-adrenergic agonists inhibit rat liver adenylate cyclase.J. Biol. Chem. 2562603–2606.
Jaspard, E., Wei, L., and Alhenc-Gelas, F. (1993). Differences in the properties and enzymatic specificities of the two active sites of angiotensin I-converting enzyme (kininase II).J. Biol. Chem. 2689496–9503.
Jeunemaire, X., Soubrier, F., Kotelevtsev, Y. V., Lifton, R. P., Williams, C. S., Charru, A., Hunt, S. C., Hopkins, P. N., Williams, R. R., Lalouel, J.-M., and Corvol, P. (1992). Molecular basis of human hypertension: role of angiotensinogen.Cell 71169–180.
Kapas, S., Hinson, J. P., Puddefoot, J. R., Ho, M. M., and Vinson, G. P. (1994). Internalization of the type I angiotensin II receptor (AT1) is required for protein kinase C activation but not for inositol trisphosphate release in the angiotensin II stimulated rat adrenal zona glomerulosa cell.Biochem. Biophys. Res. Commun. 2041292–1298.
Kata, A. M. (1992). Is angiotensin II a growth factor masquerading as a vasopressor?Heart Dis. Stroke 1151–154.
Kobayashi, S., Nishimura, J., and Kanaide, H. (1994). Cytosolic Ca2+ transients are not required for platelet-derived growth factor to induce cell cycle progression of vascular smooth muscle cells in primary culture.J. Biol. Chem. 2699011–9018.
Konishi, H., Kuroda, S., Inada, Y., and Fujisawa, Y. (1994). Novel subtype of human angiotensin II type 1 receptor: cDNA cloning and expression.Biochem. Biophys. Res. Commun. 199467–474.
Kuroda, S., Konishi, H., Okishio, M., and Fujisawa, Y. (1994). Novel subtype of human angiotensin II type 1 receptor: Analysis of signal transduction mechanism in transfected Chinese hamster ovary cells.Biochem. Biophys. Res. Commun. 199475–481.
Langford, K. G., Shai, S.-Y., Howard, T. E., Kovac, M. J., Overbeek, P. A., and Bernstein, K. E. (1991). Transgenic mice demonstrate a testis-specific promoter for angiotensin-converting enzyme.J. Biol. Chem. 26615559–15562.
Laragh, J. H., and Brenner, B. M. (eds.) (1990).Hypertension: Pathophysiology, Diagnosis and Management, Raven Press, New York.
Marsden, P. A., Brenner, B. M., and Ballermann, B. J. (1990). In Laragh, J. H., and Brenner, B. M. (eds.),Hypertension: Pathophysiology, Diagnosis, and Management, Raven Press, New York, pp. 1247–1272.
Moroianu, J., and Riordan, J. F. (1994). Nuclear translocation of angiogenic proteins in endothelial cells: An essential step in angiogenesis.Biochemistry 3312535–12539.
Mukoyama, M., Nakajima, M., Horiuchi, M., Sasamura, H., Pratt, R. E., and Dzau, V. J. (1993). Expression cloning of type 2 angiotensin II receptor reveals a unique class of seven-transmembrane receptors.J. Biol. Chem. 26824539–24542.
Nakajima, M., Mukoyama, M., Pratt, R. E., Horiuchi, M., and Dazu, V. J. (1993). Cloning of cDNA and analysis of the gene for mouse angiotensin II type 2 receptor.Biochem. Biophys. Res. Commun. 197393–399.
Neuss, M., Regitz-Zagrosek, V., Hildebrandt, A., and Fleck, E. (1994). Human cardiac fibroblasts express an angiotensin receptor with unusual binding characteristics which is coupled to cellular proliferation.Biochem. Biophys. Res. Commun. 2041334–1339.
Patchett, A. A., and Cordes, E. H. (1985). The design and properties of N-carboxyalkyldipeptide inhibitors of angiotensin-converting enzyme.Adv. Enzymol. 571–84.
Peach, M. J. (1986). Pharmacology of angiotensin II. In Fisher, A. W. (ed.),Kidney Hormones (Vol. III), Academic Press, London, pp. 274–311.
Robertson, J. I. S., and Nicholls, M. G. (eds.) (1993).The Renin-Angiotensin System (Vols. 1 and 2), Gower Medical Publishing, London.
Sasamura, H., Hein, L., Krieger, J. E., Pratt, R. E., Kobilka, B., and Dazu, V. J. (1992). Cloning, characterization and expression of two angiotensin receptor (AT-1) isoforms from the mouse genome,Biochem. Biophys. Res. Commun. 185253–259.
Schwab, D. A., and Wilson, J. E. (1989). Complete amino acid sequence of rat brain hexokinase, deduced from the cloned cDNA, and proposed structure of a mammalian hexokinase.Proc. Natl. Acad. Sci. USA 862563–2567.
Sealey, J. E., and Laragh, J. H. (1990). The renin-angiotensin-aldosterone system for normal regulation of blood pressure and sodium and potassium homeostasis. In Laragh, J. H., and Brenner, B. M. (eds.),Hypertension: Pathophysiology, Diagnosis, and Management, Raven Press, New York, pp. 1287–1310.
Shapiro, R., Holmquist, B., and Riordan, J. F. (1983). Anion activation of angiotensin converting enzyme: Dependence on nature of substrate.Biochemistry 223850–3857.
Sharma, H. S., van Heugten, H. A. A., Goedblood, M., Verdouw, P. D., and Lamers, J. M. J. (1994). Angiotensin II induced expression of transcription factors precedes increase in transforming growth factor-β1 mRNA in neonatal cardiac fibroblasts.Biochem. Biophys. Res. Commun. 205105–112.
Shigeri, Y., and Fujimoto, M. (1994). Y2 receptors for neuropeptide Y are coupled to three intracellular signal transduction pathways in a human neuroblastoma cell line.J. Biol. Chem. 2698842–8849.
Shorb, W., Peeler, T. C., Madigan, N. N., Conrad, K. M., and Baker, K. M. (1994). Angiotensin II-induced protein tyrosine phosphorylation in neonatal rat cardiac fibroblasts.J. Biol. Chem. 26919626–19632.
Skeggs, L. T., Lentz, K. E., Kahn, J. R., Shumway, N. P., and Woods, K. R. (1956). The amino acid sequence of hypertensin II.J. Exp. Med. 104193–197.
Soubrier, F., Alhenc-Gelas, F., Hubert, C., Allegrini, J., John, M., Tregear, G., and Corvol, P. (1988). Two putative active centers in human angiotensin I-converting enzyme revealed by molecular cloning.Proc. Natl. Acad. Sci. USA 859386–9350.
Stöcker, W., Wolz, R. L., Zwilling, R., Strydom, D. J., and Auld, D. S. (1988).Astacus protease, a zinc metalloenzyme.Biochemistry 275026–5032.
Tewksbury, D. A. (1990). Angiotensin: Biochemistry and molecular biology. In Laragh, J. H., and Brenner, B. M. (eds.),Hypertension: Pathophysiology, Diagnosis, and Management, Raven Press, New York.
Timmermans, P. B. M. W. M., Benfield, P., Chiu, A. T., Herblin, W. F., Benfield, P., Carini, D. J., Lee, R. J., Wexler, R. R., Saye, J. M., and Smith, R. D. (1993). Angiotensin II receptors and angiotensin II receptor antagonists.Pharmacol. Rev. 45205–251.
Urata, H., Kinoshita, A., Misono, K. S., Bumpus, F. M., and Husain, A. (1990). Identification of a highly specific chymase as the major angiotensin II-forming enzyme in the human heart.J. Biol. Chem. 26522348–22357.
Wei, L., Alhenc-Gelas, F., Corvol, P., and Clauser, E. (1991). The two homologous domains of human angiotensin I-converting enzyme are both catalytically active.J. Biol. Chem. 2669002–9008.
Wong, P. C., Price, W. A., Chiu, A. T., Duncia, J. V., Carini, D. J., Wexler, R. R., Johnson, A. L., and Timmermans, P. B. M. W. M. (1990). Nonpeptide angiotensin II receptor antagonists. VIII. Characterization of functional antagonism displayed by DuP 753, an orally active antihypertensive agent.J. Pharmacol. Exp. Ther. 252719–725.
Yang, H. Y. T., Erdös, E. G., and Levin, Y. (1970). A dipeptidyl carboxypeptidase that converts angiotensin I and inactivates bradykinin.Biochem. Biophys. Res. Commun. 214374–376.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Riordan, J.F. Angiotensin II: Biosynthesis, molecular recognition, and signal transduction. Cell Mol Neurobiol 15, 637–651 (1995). https://doi.org/10.1007/BF02071129
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02071129