Abstract
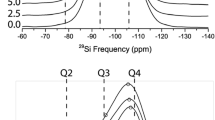
29Si-MAS NMR spectroscopy has been proved to be a versatile tool to follow the reaction development from the signal ratio of the initial material and the crystallization product, if the spectra of both compounds could be clearly distinguished in each sample. On this basis the hydrothermal transformation of kaolinite into nitrite-sodalite Na8[AlSiO4]6(NO2)2 has been investigated at a temperature of 353 K. The crystallization process was followed in the mid-stage of crystal growth for times from 6 to 24 h. The role of carbonate impurities as well as the influence of the time of nitrite-salt addition to the reaction mixture have also been included in this experimental study.
Similar content being viewed by others
References
G. Engelhardt, D. Michel:High Resolution Solid State NMR of Silicates and Zeolites. John Wiley & Sons, Chichester, New York, Brisbane 1988.
S. Hayashi, K. Suzuki, S. Shin, K. Hayamizu, O. Yamamoto:Chem. Phys. Lett. 110, 54 (1984).
J.-Ch. Buhl, C. Mundus, J. Löns, W. Hoffmann:Z. Naturforsch.,49a, 1171 (1994).
W. Borchert, J. Keidel:Heidelbg. Beitr. Mineral. Petrogr.,1, 1 (1947).
R. M. Barrer, E. A. D. White:J. Chem. Soc., 1562 (1952).
R. M. Barrer:The Hydrothermal Chemistry of Zeolites. Academic Press, London 1982.
F. Hund:Z. anorg. allg. Chem.,511, 255 (1984).
J.-Ch. Buhl:J. Cryst. Growth,108, 143 (1991).
J.-Ch. Buhl:Thermochim. Acta,219, 205 (1993).
G. Engelhardt, S. Luger, J.-Ch. Buhl, J. Felsche:Zeolites,9, 182 (1989).
C. Mundus: PhD thesis, Phys. Chem. Institute University of Münster, Münster, 1994.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Buhl, JC., Reich, D., Mundus, C. et al. 29Si MAS NMR investigations on the crystallization of nitrite-sodalite. React Kinet Catal Lett 58, 13–18 (1996). https://doi.org/10.1007/BF02071099
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02071099