Abstract
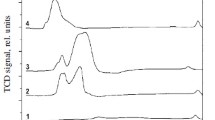
Catalytic properties of copper intermetallides with Mg, Ga, In, La, Pr, Si, Ti, Ge, Zr, Sn, Se and Te have been examined. Most selective with respect to the yield of formaldehyde are Cu3Si and Cu31Sn8. Cu9Ga4, Cu3Ge, Cu2Se and Cu2Te are catalytically inactive. Cu6La and Cu6Pr possess high dehydrogenating activity but are not selective toward formaldehyde.
Abstract
Исследованы каталитические свойства интерметаллидов меди с Mg, Ga, In, La, Pr, Si, Ge, Zr, Sn, Se, Te. Наиболее избирательны в отношении выхода формальдегида Cu3Si и Cu31Sn8. Cu9Ga4, Cu3Ge, Cu2Se и Cu2Te каталитически не активны. Cu6La и Cu6Pr облададают сильной дегидрирующей способностью, но не избирательны в отношении выхода формальдегида.
Similar content being viewed by others
References
V. S. Kovalenko: Metallographic Reactants, Metallurgia, Moskva 1970.
B. I. Popov, N. G. Skomorokhova: React. Kinet. Catal. Lett.,25, (1984) (in press).
M. Hansen, K. Anderko: Double Alloy Structures, vol. 2, Metallurgia, Moskva 1962.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Papov, B.I., Skomorokhova, N.G., Karonik, V.V. et al. Catalytic properties of some copper intermetallides in the oxidative dehydrogenation of methanol. React Kinet Catal Lett 27, 419–423 (1985). https://doi.org/10.1007/BF02070486
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02070486