Abstract
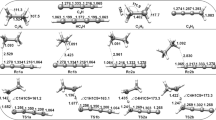
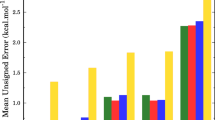
The transition states of intramolecular 1,4 and 1,5 H-atom transfers, from/to primary (p), secondary (s) or tertiary alkyl (t) and primary (p a), secondary (s a) or tertiary (t a) allyl carbon atoms, have been studied at the level of the semiempirical quantum-chemical method AM1 with the UHF approximation. The activation and reaction enthalpies were calculated and compared with experimental data available in the literature and the calculated data obtained for analogous reactions in alkyl radicals. Correlations were found between the activation enthalpies and the dissociation enthalpies of the bonds broken and formed.
Similar content being viewed by others
References
J. W. Wilt: inFree Radicals, Chap. 8, Vol.1, J.K. Kochi, Ed. Wiley-Interscience, New York 1978.
S. Dóbé, T. Bérces: inChemical Kinetics of Small Organic Radicals, Ed. Z.B. Alfassi, Vol. 1, p. 89, CRC Press, Boca Raton, Florida 1988.
S. Dóbé, T. Bérces, F. Márta:Int. J. Chem. Kinet. 19, 895 (1987).
K.W. Watkins, D.R. Lawson:J. Phys. Chem. 75, 1632 (1971).
L. Seres, R. Fischer, K. Scherzer, M. Görgényi:J.C.S. Faraday Trans.,91, 1303 (1995).
A.L.J. Beckwith, K.U. Ingold: inRearrangements in Ground and Excited States, Vol. 1, P. de Mayo, Ed. Academic Press, New York 1980.
A.L.J. Beckwith:Tetrahedron,37, 3073 (1981).
T. Körtvélyesi, L. Seres:React. Kinet. Catal. Lett.,58, 27 (1996).
B. Viskolcz, Gy. Lendvay, T.Körtvélyesi, L. Seres:J. Am. Chem. Soc., to be published.
M.J.S. Dewar, E.G. Zoebish, E.F. Healy, J.J.P. Stewart:J. Am. Chem. Soc.,107, 3902 (1985).
J.J.P. Stewart: MOPAC Version 6.1, 1990.
S.W. Benson:Thermochemical Kinetics, 2nd ed. Wiley-Interscience, New York 1976.
T. Bérces, Z. Szabó:MTA Kémiai Oszt. Közl.,19, 303 (1963).
M.G. Evans, M. Polanyi:Trans. Faraday Soc.,32, 1333 (1956).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Körtvélyesi, T., Seres, L. UHF-AM1 studies on 1,4 and 1,5 intramolecular H-atom transfer reactions in alkenyl radicals. React Kinet Catal Lett 59, 35–40 (1996). https://doi.org/10.1007/BF02067989
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02067989