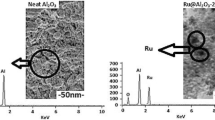
Abstract
The conversion of a carboxylic acid was increased to 90% with 100% selectivity to the corresponding alcohol by calcination of the catalyst precursor, Sn/alumina, of Ru−Sn/alumina in the hydrogenation of the carboxylic acid.
Similar content being viewed by others
References
O.A. Ferreti, J.P. Bournouville, G. Mabilon, G. Martino, J.P. Candy, J.M. Basset:J. Mol. Catal.,67, 283 (1991).
C.S. Narasimhan, V.M. Deshpande, K. Ramnarayan:Appl. Catal.,48, L1 (1989).
K. Tahara, Y. Itoi, S. Nishiyama, S. Tsuruya, M. Masai:Nippon Kagaku Kaishi, 100–106, 1995.
K.Y. Cheah, T.S. Tang, F. Mizukami, S. Niwa, M. Toba, Y.M. Choo:J. Am. Oil. Chem. Soc.,69, 410 (1992).
K. Tahara, E. Nagahara, Y. Itoi, S. Nishiyama, S. Tsuruya, M. Masai:74th CATSJ Meeting Abstracts: No. 3F02 (1994).
The acid value and the hydroxy value are defined byJapanese Industrial Standard K0070.
H. Verbeek, W.M.H. Sachtler:J. Catal.,42, 257 (1976).
R. Bouwman, L.H. Toneman, A.A. Holscher:Surf. Sci.,35, 8 (1973).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tahara, K., Nagahara, E., Itoi, Y. et al. Effect of calcination temperature during preparation of Ru−Sn-alumina catalysts on the hydrogenation of carboxylic acid. React Kinet Catal Lett 59, 15–18 (1996). https://doi.org/10.1007/BF02067986
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02067986