Abstract
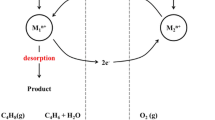
Catalytic activity of ferric oxide was evaluated in the oxidative dehydrogenation of n-butane. The apparent activation energy determined in the kinetic region was 37.7 kcal/mol. The activity of this oxide was undetectable up to 430°C. Reaction product distributions are shown as functions of temperature in the range of 430–650°C.
Abstract
Каталитическую активность окиси железа (III) измеряли в окислительном дегидрировании н-бутана. Кажущаяся энергия активации равна 37,7 ккал/моль. Активность окисла не удалось обнаружить вплоть до 430°C. Распределение продуктов реакции представлено в зависимости от температуры в интервале 430–650°C.
Article PDF
Similar content being viewed by others
References
L.C. Romero, E.E. Gonzo, J.C. Gottifredi: React. Kinet. Catal. Lett., in press.
E.E. Gonzo, L.C. Romero, J.C. Gottifredi: React. Kinet. Catal. Lett., in press.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Romero, L.C., Gonzo, E.E. & Gottifredi, J.C. Oxidative dehydrogenation of n-butane, III. Kinetic and catalytic activity of ferric oxide. React Kinet Catal Lett 38, 375–379 (1989). https://doi.org/10.1007/BF02062134
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02062134