Abstract
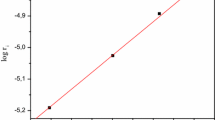
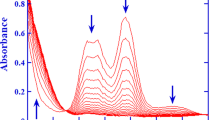
Kinetics of H2 evolution in the RhCl3−NaH2PO2−HCl−H2O system has been studied. As found by31P NMR, Rh(I) complexes with the Rh−P(OH)2O− bond are formed and play the role of catalysts in the subsequent H2 evolution reaction.
Abstract
Проведено исследование кинетики выделения H2 в системе RhCl3−NaH2PO2−HCl−H2O. Методом ЯМР31P обнаружено образование комплексов Rh(I) со связью Rh−P(OH)2O−, играющих роль катализаторов в последующем выделении H2.
Similar content being viewed by others
References
Ya. I. Valsyunene, A. Yu. Yuryavichus: Zh. Neorg. Khim.,29, 2600 (1984).
E. N. Yurchenko, N. P. Anikeenko: React. Kinet. Catal. Lett.,2, 65 (1975).
E. N. Yurchenko: Kinet. Katal.,14, 515 (1973).
E. N. Yurchenko, A. D. Troitskaya, L. S. Gracheva, L. D. Alt: Izv. AN SSSR, Ser. Khim.,10, 215 (1975).
E. N. Yurchenko, L. S. Gracheva, L. Ya. Alt, L. A. Pavlyakhina: Koord. Khim.,7, 930 (1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yurchenko, E.N., Yuryavichus, A.Y., Fedotov, M.A. et al. Catalytic oxidative hydrolysis of H2PO −2 anions in the presence of RhCl3 . React Kinet Catal Lett 39, 55–60 (1989). https://doi.org/10.1007/BF02061855
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02061855