Abstract
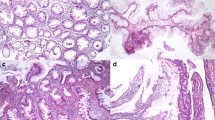
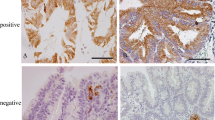
The transitional mucosa (TM) adjacent to carcinomas of the large bowel shows histologic and mucin histochemical changes that may indicate premalignant change and may be of prognostic value after radical resection. In this study, 10 anterior resection specimens from patients with carcinomas of the rectum and rectosigmoid were used to compare the nuclear DNA content in TM with those in cancer tissue and with those in nontransitional mucosa (N-TM;i.e.,uninvolved mucosa remote from tumors showing normal histologic and mucin histochemical features). The nuclear DNA content was assessed using DNA image cytometry on Feulgen-stained sections. As compared with N-TM, crypts in TM contained greater numbers of cells, were elongated, and were more likely to be branched with marked sialomucin secretion, accompanied by a marked reduction in the normal sulfomucin content. The mean nuclear DNA content in the upper, middle, and lower thirds of crypts was significantly higher in TM than in N-TM, and the nuclear DNA content in TM exhibited no correlation to that in tumors. The results suggest that TM adjacent to carcinomas of the rectum and rectosigmoid has higher proliferative activity, providing further evidence that TM may be an unstable premalignant change.
Similar content being viewed by others
References
Filipe MI. Value of histochemical reactions for mucosubstances in the diagnosis of certain pathological conditions of the colon and rectum. Gut 1969;10:577–86.
Filipe MI, Branfoot AC. Abnormal patterns of mucus secretion in apparently normal mucosa of large intestine with carcinoma. Cancer 1974;34:282–90.
Wang Q, Gao H, Wang Y, Chen Y. The clinical and biological significance of the transitional mucosa adjacent to colorectal cancer. Jpn J Surg 1991;21:253–61.
Graves P, Filipe MI, Branfoot AC. Transitional mucosa and survival in human colorectal cancer. Cancer 1980;46:764–70.
Habib NA, Dawson PM, Blount MA, Cox S, Krausz T, Wood CB. Study of the histochemical changes in mucus from normal and tumour bearing mucosa in patients with colorectal cancer. Eur J Surg Oncol 1985;11:243–5.
Dawson PM, Habib NA, Rees HC, Wood CB. Mucosal field change in colorectal cancer. Am J Surg 1987;153:281–4.
Wang Q, Wang FM, Gao H, Chen YL, Wang YH, He J. Sialomucin at the resection margins and likelihood of local recurrence of carcinoma of the rectum after anterior resection. Ann Med 1990;22:31–3.
Andreola S, Bertario L, Bombelli L, Rossetti C, Gennari L. Abnormal pattern of mucin secretion at the resection margin of colorectal carcinomas. Tumori 1988;74:317–20.
Dawson PM, Habib NA, Rees HC, Williamson RC, Wood CB. Influence of sialomucin at the resection margin on local tumour recurrence and survival of patients with colorectal cancer: a multivariate analysis. Br J Surg 1987;74:366–9.
Coloacchio TA, Dressel D, Dunn JL. Efficacy of differential mucin staining for predicting synchronous and metachronous colorectal carcinomas. Am J Surg 1987;153:144–8.
Williams GT. Transitional mucosa of the large intestine. Histopathology 1985;9:1237–43.
Turnbull RB Jr, Kyle K, Watson FR, Spratt J. Cancer of the colon: the influence of theno-touch isolation technique on survival rates. Ann Surg 1967;166:420–7.
Bara J, André J, Gautier R, Burtin P. Abnormal pattern of mucus-associated Ml antigens in histologically normal mucosa adjacent to colonic adenocarcinomas. Cancer Res 1984;44:4040–5.
Filipe MI, Lake BD, eds. Histochemistry in pathology. Appendix 3. Carbohydrates, mucins, mucopolysaccharides. Edinburgh, New York: Churchill Livingstone, 1983:310–7.
Mays RG. The Feulgen hydrolysis profiles a metastatic lung tumour in lymph node and a primary breast carcinoma. Med Lab Sci 1987;44:307–11.
Allen DC, Connolly NS, Biggart JD. High iron diamine-alcian blue mucin profiles in benign, premalignant and malignant colorectal disease. Histopathology 1988;13:399–411.
Schwarz H. Morphometric requirements for image analysis and the IBAS interactive automatic image analysis system. Anal Quant Cytol Histol 1986;8:267–70.
Saffos RO, Rhatigan RM. Benign (nonpolypoid) mucosal changes adjacent to carcinomas of the colon: a light microscopic study of 20 cases. Hum Pathol 1977;8:441–9.
Hamilton PW, Watt PC, Allen DC. A morphometric assessment of transitional mucosa in the colon. Histopathology 1988;13:519–30.
Dawson PA, Filipe MI. An ultrastructural and histochemical study of the mucous membrane adjacent to and remote from carcinoma of the colon. Cancer 1976;37:2388–98.
Riddell RH, Levin B. Ultrastructure of the “transitional” mucosa adjacent to large bowel carcinoma. Cancer 1977;40:2509–22.
Dawson PA, Filipe MI. An ultrastructural application of silver methenamine to the study of mucin changes in the colonic mucosa adjacent to and remote from carcinoma. Histochem J 1976;8:143–58.
Suzuki H, Umehara N. Early mucosal changes in dimethylhydrazine-induced colonic carcinogenesis in rats. Jpn J Surg 1986;16:140–3.
Matthews J, Cooke T. Changes in crypt cell DNA content during experimental colonic carcinogenesis. Br J Cancer 1986;53:787–91.
Lev R. A histochemical study of the glycogen and mucin in developing human foetal epithelia. Histochem J 1968;1:152–65.
Caccamo D, Telenta M, Celener D. Concanavalin A binding sites in fetal, adult, transitional, and malignant rectosigmoid mucosa. Hum Pathol 1989;20:1186–92.
Filipe MI. Mucous secretion in rat colonic mucosa during carcinogenesis induced by dimethylhydrazine: a morphological and histochemical study. Br J Cancer 1975;32:60–77.
Filipe MI, Scurr JH, Ellis H. Effects of fecal stream on experimental colorectal carcinogenesis: morphologic and histochemical changes. Cancer 1982;50:2859–65.
Isaacson P, Attwood PR. Failure to demonstrate specificity of the morphological and histochemical changes in mucosa adjacent to colonic carcinoma (transitional mucosa). J Clin Pathol 1979;32:214–8.
Listinsky CM, Riddell RH. Patterns of mucin secretion in neoplastic and non-neoplastic diseases of the colon. Hum Pathol 1981;12:923–9.
Mori M, Shimono R, Adachi Y,et al. Transitional mucosa in human colorectal lesions. Dis Colon Rectum 1990;33:498–501.
Bara J, Burtin P. Mucous-associated gastrointestinal antigens in transitional mucosa adjacent to human colonic adenocarcinomas: their ‘fetal type’ association. Eur J Cancer 1980;16:1303–10.
Wang Q, Gao H, Wang FM,et al. The immunohistochemical study of carcinoembryonic antigen in the transitional mucosa adjacent to colorectal carcinomas. Zhong Hua Xiaohua Zazhi (Chin J Dig) 1990;10:25–7.
Lapertosa G, Baracchini P, Abbas S, Fulcheri E, Tanzi R, Filipe MI. Tissue evaluation of epithelial and functional markers of cell differentiation and mucins in colonic malignancy: assessment of diagnostic and prognostic value. Pathologica 1988;80:145–57.
Shi ZR, McIntyre LJ, Knowles BB, Solter D, Kim YS. Expression of a carbohydrate differentiation antigen, stage-specific embryonic antigen 1, in human co-Ionic adenocarcinoma. Cancer Res 1984;44:1142–7.
Xu M, Real FX, Welt S, Schüssler MH, Oettgen HF, Old LJ. Expression of TAG-72 in normal colon, transitional mucosa, and colon cancer. Int J Cancer 1989;44:985–9.
Boland CR, Montgomery CK, Kim YS. Alterations in human colonic mucin occurring with cellular differentiation and malignant transformation. Proc Natl Acad Sci U S A 1982;79:2051–5.
Cooper HS. Peanut lectin-binding sites in large bowel carcinomas. Lab Invest 1982;47:383–90.
Calderó J, Campo E, Ascaso C, Ramos J, Panadés MJ, René JM. Regional distribution of glycoconjugates in normal, transitional and neoplastic human colonic mucosa: a histochemical study using lectins. Virchows Arch [A] 1989;415:347–56.
Author information
Authors and Affiliations
Additional information
Read at the Third Chinese-Japan Symposium on Digestive Surgery, Hangzhou, People's Republic of China, March 24 to 26, 1991.
About this article
Cite this article
Wang, Q., Gao, H., Chen, Y. et al. Biopathologic characteristics of DNA content in crypt cells of transitional mucosa adjacent to carcinomas of the rectum and rectosigmoid. Dis Colon Rectum 35, 670–675 (1992). https://doi.org/10.1007/BF02053758
Issue Date:
DOI: https://doi.org/10.1007/BF02053758