Abstract
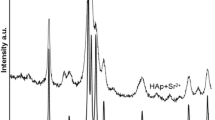
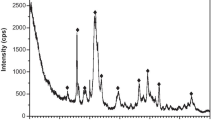
Adsorption of strontium ions on synthetic hydroxyapatite was examined using both batch and column methods. The apatite was prepared from aqueous solutions and characterized by standard analytical methods. The sample obtained had characteristics of well crystallized stoichiometric hydroxyapatite. The experimental data for sorption of strontium can be very well fitted with Langmuir's adsorption isotherm. It was found that sorption occurs by an ion exchange reaction between strontium ions in solution and calcium ions in apatite.
Similar content being viewed by others
References
J. D. TERMINE, A. S. POSNER, Science, 153 (1966) 1523.
R. ROY, Radioactive Waste Disposal, Vol. 1, Pergamon Press, New York, 1982.
IAEA Technical Reports Series No 236, Vienna, 1984, p. 51.
R. W. LEGGETT, K. F. ECKERMAN, L. R. WILLIAMS, Health Phys., 3 (1982) 307.
B. TORSTENFELT, K. ANDERSSON, B. ALLARD, Chem. Geol., 36 (1982) No. 1-2, 123.
R. A. LAUDERDALE, Ind. Eng. Chem., 43 (1951) 1538.
V. P. SHVEDOV et al., Radiokhimiya, 8 (1966) 369.
PH. GRAMAIN, J. C. VOEGEL, M. GUMPPER, J. H. THOMANN, J. Colloid Interface Sci., 118 (1987) 148.
R. M. H. VERBEEK, H. STEYAER, H. P. THUN, F. VERBEEK, J. Chem. Soc. Faraday Trans. I, 76 (1980) 209.
H. H. ROOTARE, V. R. DEITZ, F. G. CARPENTER, J. Colloid Sci., 17 (1962) 179.
H. McDOWELL, T. M. GREGORY, W. E. BROWN, J. Res. Nat. Bur. Stand., 81A (1977) 273.
H. A. FLASCHKA, EDTA Titrations, Pergamon Press, London 1959, p. 77.
J. MURPHY, J. P. RILEY, Anal. Chim. Acta, 27 (1962) 21.
W. G. PERDOK, J. CHRISTOFFERSEN, J. ARENDS, J. Crystal Growth, 80 (1987) 149.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lazić, S., Vuković, Ž. Ion exchange of strontium on synthetic hydroxyapatite. Journal of Radioanalytical and Nuclear Chemistry, Articles 149, 161–168 (1991). https://doi.org/10.1007/BF02053724
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02053724