Abstract
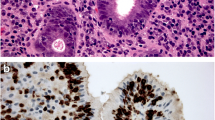
PURPOSE: Expression of disaccharidase sucrase-isomaltase (SI) is significantly enhanced during neoplastic transformation of colonic epithelium. Our study was designed to determine whether expression of SI within primary tumors was significantly associated with survival in patients with colorectal carcinoma (CRC). METHODS: SI expression was analyzed by immunohistochemistry in paraffin sections from 182 Stage I to III CRC that had been resected for cure at the New England Deaconess Hospital between 1965 and 1977. Expression was scored as absent or present in 1 to 50 percent or more than 50 percent of tumor cells. Associations were explored among SI expression, other clinical or pathologic variables, and overall survival. The data set is mature, with 91 (56 percent) patients who had died of CRC at a median follow-up of 96 months. RESULTS: Fifty-five percent of primary CRC expressed SI. When the multivariate Cox analysis was performed, nodal status, T stage, primary site, grade, and SI expression were independent covariates. SI expression was not associated with the expression of other clinicopathologic variables but increased the risk of death from colorectal carcinoma by 1.83-fold. DISCUSSION: These results indicate that SI is a prognostic marker for CRC that is independent of stage-related variables in patients who have undergone potentially curative resections.
Similar content being viewed by others
References
Hermanek P, Sobin LH, eds. TNM: classification of malignant tumours. 4th ed. Berlin: Springer-Verlag, 1987:47–50.
Jessup JM, Steele G Jr, Thomas P,et al. Molecular biology of neoplastic transformation of the large bowel: identification of two etiologic pathways. Surg Oncol Clin North Am 1994;3:449–77.
Vogelstein B, Fearon ER, Hamilton SR,et al. Genetic alterations during colorectal-tumor development. N Engl J Med 1988;319:525–32.
Fishel R, Lescoe MK, Rao MR,et al. The human mutator gene homolog MSH2 and its association with hereditary nonpolyposis colon cancer. Cell 1993;75:1027–38.
Leach FS, Nicolaides NC, Papadopoulos N,et al. Mutations of a mutS homolog in hereditary nonpolyposis colorectal cancer. Cell 1993;75:1215–25.
Aaltonen LA, Peltomaki P, Leach FS,et al. Clues to the pathogenesis of familial colorectal cancer. Science 1993;260:812–6.
Peltomaki P, Aaltonen LA, Sistonen P,et al, Genetic mapping of a locus predisposing to human colorectal cancer. Science 1993;260:810–2.
Jen J, Kim H, Piantadosi S,et al. Allelic loss of chromosome 18q and prognosis in colorectal cancer. N Engl J Med 1994;331:213–21.
Thibodeau SN, Bren G, Schaid D. Microsatellite instability in cancer of the proximal colon. Science 1993;260:816–9.
Wiltz O, O'Hara CJ, Steele GD, Mercurio AM. Sucraseisomaltase: a marker associated with the progression of adenomatous polyps to adenocarcinomas. Surgery 1990;108:269–76.
Wiltz O, O'Hara CJ, Steele GD, Mercurio AM. Expression of enzymatically active sucrase-isomaltase is a ubiquitous property of colon adenocarcinomas. Gastroenterology 1991;200:1266–78.
Wu GD, Beer DG, Moore JH, Orringer MB, Appelman HD, Traber PG. Sucrase-isomaltase gene expression in Barrett's esophagus and adenocarcinoma. Gastroenterology 1993;105:837–44.
Nikulasson S, Andrews CW Jr, Goldman H,et al. Sucrase-isomaltase expression in dysplasia associated with Barrett's esophagus and chronic gastritis. Int J Surg Pathol (in press).
Hauri H-P, Sander B, Nairn H. Induction of lactase biosynthesis in the human intestinal epithelial cell line Caco-2. Eur J Biochem 1994;219:539–46.
Halline AG, Davidson NO, Skarosi SF,et al. Effects of 1,25-dihydroxyvitamin D3 on proliferation and differentiation of Caco-2 cells. Endocrinology 1994;134:1710–7.
Minsky BD, Mies C, Recht A, Rich TA, Chaffey JT. Resectable adenocarcinoma of the rectosigmoid and rectum: II. The influence of blood vessel invasion. Cancer 1988;61:1417–24.
Minsky BD, Mies C, Rich TA, Recht A, Chaffey JT. Potentially curative surgery of colon cancer: the influence of blood vessel invasion. J Clin Oncol 1988;6:119–27.
Minsky BD, Mies C, Rich TA, Recht A, Chaffey JT. Colloid carcinoma of the colon and rectum. Cancer 1987;60:3103–12.
Lloyd ML, Olsen WA. A study of the molecular pathology of sucrase-isomaltase deficiency: a defect in the intracellular processing of the enzyme. N Engl J Med 1987;316:438–42.
Andrews Jr CW, O'Hara CJ, Goldman H, Mercurio AM, Silverman ML, Steele Jr GD. Sucrase-isomaltase expression in chronic ulcerative colitis dysplasia. Hum Pathol 1992;23:774–9.
Loda M, Lipman J, Cukor B, Bur M, Kwan P, DeLellis RA. Nodular foci in parathyroid adenomas and hyperplasias: an immunohistochemical analysis of proliferative activity. Hum Pathol 1994;25:1050–6.
Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc 1958;53:457–81.
Gehan E. A generalized Wilcoxon test for comparing arbitrarily singly-censored data. Biometrika 1965;52:203–23.
Cox DR. Regression models and life tables (with discussion). J R Stat Soc B 1972;34:187–220.
Jonckheere AR. A distribution-free K-sample test against ordered alternatives. Biometrika 1954;41:133–45.
Real FX, Xu M, Vila MR, de Bolos C. Intestinal brush-border-associated enzymes: co-ordinated expression in colorectal cancer. Int J Cancer 1992;51:173–81.
Beaulieu J-F, Weiser MM, Herrera L, Quaroni A. Detection and characterization of sucrase-isomaltase in adult human colon and in colonic polyps. Gastroenterology 1990;98:1467–77.
Chantret I, Rodolosse A, Barbat A,et al. Differential expression of sucrase-isomaltase in clones isolated from early and late passages of the cell line Caco-2: evidence for glucose-dependent negative regulation. J Cell Sci 1994;107:213–25.
Darmoul D, Baricault L, Sapin C, Chantret I, Trugnan G, Rousset M. Decrease of mRNA levels and biosynthesis of sucrase-isomaltase but not dipeptidylpeptidase IV in forskolin or monensin-treated Caco-2 cells. Experientia 1991;47:1211–5.
Zweibaum A, Pinto M, Chevalier G,et al. Enterocytic differentiation of a subpopulation of the human colon tumor cell line HT-29 selected for growth in sugar-free medium and its inhibition by glucose. J Cell Physiol 1985;122:21–9.
Chantret I, Lacasa M, Chevalier G,et al. Sequence of the complete cDNA and the 5′ structure of the human sucrase-isomaltase gene: possible homology with a yeast glucoamylase. Biochem J 1992;285:915–23.
Wu GD, Wang W, Traber PG. Isolation and characterization of the human sucrase-isomaltase gene and demonstration of intestine-specific transcriptional elements. J Biol Chem 1992;267:7863–70.
Wu GD, Chen L, Forslund K, Traber PG. Hepatocyte nuclear factor-1 alpha (HNF-1 alpha) and HNF-1 beta regulate transcription via two elements in an intestinespecific promoter. J Biol Chem 1994;269:17080–5.
Jass JR, Mukawa K, Goh HS, Love SB, Capellaro D. Clinical importance of DNA content in rectal cancer measured by flow cytometry. J Clin Pathol 1989;42:254–9.
Kouri M, Pyrhonen S, Mecklin JP,et al. The prognostic value of DNA-ploidy in colorectal carcinoma: a prospective study. Br J Cancer 1990;62:976–81.
Fearon ER, Cho KR, Nigro JM,et al. Identification of a chromosome 18q gene that is altered in colorectal cancers. Science 1990;247:49–56.
Minsky BD, Mies C, Rich TA, Recht A. Lymphatic vessel invasion is an independent prognostic factor for survival in colorectal cancer. Int J Radiat Oncol Biol Phys 1989;17:311–8.
Author information
Authors and Affiliations
Additional information
Supported by Grant CA44704 from the National Institutes of Health, Department of Health and Human Services, Washington, D.C. Read at the meeting of The American Society of Colon and Rectal Surgeons, Montreal, Quebec, Canada, May 7 to 12, 1995.
About this article
Cite this article
Jessup, J.M., Lavin, P.T., Andrews, C.W. et al. Sucrase-isomaltase is an independent prognostic marker for colorectal carcinoma. Dis Colon Rectum 38, 1257–1264 (1995). https://doi.org/10.1007/BF02049149
Issue Date:
DOI: https://doi.org/10.1007/BF02049149