Abstract
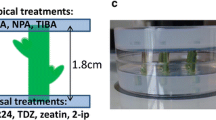
The stylar abscission bioassay was used to identify five stimulators of lemon (Citrus limon cv. Lisbon) abscission in pistil explants. The stimulators (N-phenyl-N′-1,2,3-thiadiazol-5-ylurea, N6-benzyladenine, kinetin, zeatin, and N6-isopentenyladenine), which are all cytokinins, accelerated the timing of expiant abscission when they were added as supplements (100 μM) to the test medium. To study possible relationships between cytokinins, ethylene, and abscission, we measured accumulating ethylene concentrations in sealed cultures and endogenous 1 -aminocyclopropane-1-carboxylic aicd (ACC) in explants incubated on test medium plus or minus 100 μM N-phenyl-N′-1,2,3-thiadiazol-5-ylurea (thidiazuron), 100 μM N6-benzyladenine (bzl6Ade), or 2 μM picloram, an inhibitor of stylar abscission. Although ethylene accumulated to similar levels in all treatments, the concentrations obtained with picloram and thidiazuron were, respectively, higher and lower than those obtained in control cultures. The accumulation of ethylene in cultures with bzl6Ade, on the other hand, was not significantly different from controls. ACC concentrations in explants remained fairly constant in all treatments during the incubations, except in explants on thidiazuron, in which case the ACC concentration declined slightly. We conclude that cytokinins can stimulateCitrus abscissionin vitro and that this stimulation is not accompanied by marked effects on either measurable ethylene or ACC concentrations. Our finding that 100 μM aminoethoxyvinylglycine, an ethylene biosynthesis inhibitor, counteracts the stimulation of abscission by bzl6Ade suggests that a minimum level of ethylene production is required for the cytokinin effect. The possibility that cytokinins affect other aspects related to ethylene, such as biosynthetic rates, metabolism, or tissue retention, is not excluded by our results.
Similar content being viewed by others
Abbreviations
- ABA:
-
abscisic acid
- ACC:
-
1-aminocyclopropane-1-carboxylic acid
- bzl6Ade:
-
N6-benzyladenine
- io6Ade:
-
zeatin
- i6Ade:
-
N6-isopentenyladenine
- AVG:
-
aminoethoxyvinylglycine
References
Addicott FT (1982) Abscission. University of California Press, Berkeley
Beyer EM Jr, Blomstrom DC (1980) Ethylene metabolism and its possible physiological role in plants. In: Skoog F (ed) Plant growth substances 1979. Springer-Verlag, Berlin Heidelberg New York, pp 208–218
Boller T, Herner RC, Kende H (1979) Assay and enzymatic formation of an ethylene precursor, 1-aminocyclopropane-1-carboxylic acid. Planta 145:293–303
Carr DJ, Burrows WJ (1967) Studies on leaflet abscission of blue lupine leaves. I. Interaction of leaf age, kinetin, and light. Planta 73:357–368
Chatterjee SK, Leopold AC (1964) Kinetin and gibberellin actions on abscission processes. Plant Physiol 39:334–337
Einset JW, Cheng A, Elhag H (1980) Citrus tissue culture: Regulation of stylar abscission in excised pistils. Can J Bot 58:1257–1261
Einset JW, Lyon JL, Johnson P (1981a) Chemical control of abscission and degreening in stored lemons. J Amer Soc Hort Sci 106:531–533
Einset JW, Lyon JL, Sipes DL (1981b) Citrus tissue culture: Auxins in relation to abscission in excised pistils. Plant Physiol 67:1109–1112
Goldschmidt EE, Leshem Y (1971) Style abscission in the citron (Citrus medica L.) and otherCitrus species: Morphology, physiology, and chemical control with picloram. Amer J Bot 58:14–23
Hall MA, Cairns AJ, Evans DE, Smith AR, Smith PG, Taylor JE, Thomas CJR (1982) Binding sites for ethylene. In: Wareing PF (ed) Plant growth substances 1982. Academic Press, London, pp 375–383
Lau OL, John WW, Yang SF (1977) Effect of different cytokinins on ethylene production by mung bean hypocotyls in the presence of indole-3-acetic acid or calcium ion. Physiol Plant 39:1–3
Lewis LN, Palmer RL, Hield HZ (1968) Interactions of auxins, abscission accelerators, and ethylene in the abscission of citrus fruit. In: Wightman F, Setterfield G (eds) Biochemistry and physiology of plant growth substances. Runge Press, Ottawa, pp 1303–1313
Lieberman M (1979) Biosynthesis and action of ethylene. Ann Rev Plant Physiol 30:533–591
Lizada MCC, Yang SF (1979) A simple and sensitive assay for 1-aminocyclopropane-1-carboxylic acid. Anal Biochem 100:140–145
Mok MC, Mok DWS, Armstrong DJ, Shudo K, Isogai Y, Okamoto T (1982) Cytokinin activity of N-phenyl-N′-1,2,3-thiadiazol-5-ylurea (thidiazuron). Phytochemistry 21:1509–1511
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Nauer EM, Boswell SB, Holmes RC (1979) Chemical treatments, greenhouse temperatures, and supplemental day length effect forcing and growth of newly budded orange trees. HortScience 14:229–231
Pierik RLM (1980) Hormonal regulation of secondary abscission in pear pedicelsin vitro. Physiol Plant 48:5–8
Sagee O, Goren R, Riov J (1980) Abscission of citrus leaf explants: Interrelationships of abscisic acid, ethylene, and hydrolytic enzymes. Plant Physiol 66:750–753
Sipes DL, Einset JW (1982) Role of ethylene in stimulating stylar abscission in pistil explants of lemons. Physiol Plant 56:6–10
Yang SF, Adams DO, Lizada C, Yu Y, Bradford KJ, Cameron AC, Hoffman NE (1980) Mechanism and regulation of ethylene biosynthesis. In: Skoog F (ed) Plant growth substances 1979. Springer-Verlag, Berlin Heidelberg New York, pp 219–229
Yang SF, Hoffman NE, McKeon T, Riov J, Kao CH, Yung KH (1982) Mechanism and regulation of ethylene biosynthesis. In: Wareing PF (ed) Plant growth substances 1982. Academic Press, London, pp 239–248.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sipes, D.L., Einset, J.W. Cytokinin stimulation of abscission in lemon pistil explants. J Plant Growth Regul 2, 73–80 (1983). https://doi.org/10.1007/BF02042235
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02042235