Abstract
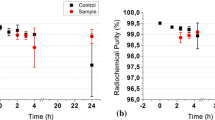
The kinetics of99mTc-d, 1-HMPAO decomposition is studied using home kits. The results showed that99mTc-d, 1-HMPAO decomposition is a first-order reaction. The decomposition constant k is found to be 0.017±0.007h−1 under the experimental conditions of 20°C, 185MBq/ml, pH 7.0. The stability of99mTc-d, 1-HMPAO is affected not only by pH and radioactive concentration, but also by temperature. Using Immol/l gentisic acid as a stabilizer, 740MBq/ml of99mTc-d, 1-HMPAO can be stabilized for 3h with the radiochemical purity above 80%.
Similar content being viewed by others
References
R. D. NEIRINCKX, L. R. CANNING, I. M. PIPER, J. Nucl. Med., 28 (1987) 191.
C. H. KAO, K. S. HSIEH, Y. L. WANG Nucl. Med. Commun., 12 (1991) 951.
N. V. RAZUMENIC, N. PUJIC, N. DEDOVIC, Nucl. Med. Biol., 19 (1992) 251.
V. J. EYNE, A. FORSTER, D. A. TYRRELL, Nucl. Med. Commun., 10 (1989) 29.
J. C. HUNG, C. MELVIN, W. A. VOLKERT, Nucl. Med., 29 (1988) 1568.
J. C. HUNG, W. A. VOLKERT, R. A. HOLMES, Nucl. Med. Biol., 16 (1989) 675.
S. JURISSON, E. O. SCHLEMPER, D. E. TROUTNER, Inorg. Chem., 25 (1986) 543.
K. TUBERGEN, M. CORLIJA, W. A. VOLKERT, J. Nucl. Med., 32 (1991) 111.
A. TOFE, J. BEVAN, M. B. FAWZI, J. Nucl. Med., 21 (1980) 366.
J. LANG, E. BATBARICS, J. LAZAR, Eur. J. Nucl. Med., 16 (1989) 424.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jinming, Z., Jiahe, T., Qiongfang, L. et al. Kinetic analysis of99mTc-d, 1-HMPAO decomposition and the method of stabilization. Journal of Radioanalytical and Nuclear Chemistry, Articles 206, 85–89 (1996). https://doi.org/10.1007/BF02040045
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02040045