Abstract
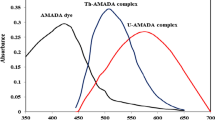
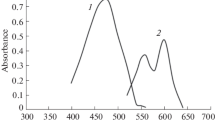
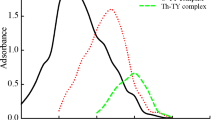
Uranium(VI) reacts with 1-(2′-thiazolylazo)-2-naphthol to form a red-coloured chelate in the pH range 5.3–7.2, maintained by 0.04 M acetate buffer. Absorbance of the sparingly soluble complex, solubilized and stabilized by Triton X-100, is measured after 30 min and it is stable for at least 16 hours. The complex exhibits maximum absorbance at 575 and 625–630 nm, but absorbance at longer wavelengths is not stable. The 1∶2 complex obeys Beer's law over the concentration range 0.4–6.4 μg of uranium(VI) per cm3, has molar absorptivity 3.36·104 dm3·mol−1·cm−1, Sandell sensitivity 7.0 ng·cm−2, formation constant (log K) 9.32 and coefficient of variation ±0.77%. Effect of 60 ions has been studied and selectivity improved considerably in presence of CDTA. The method has been applied for determination of uranium content in a rock sample.
Similar content being viewed by others
References
L. SOMMER, V. M. IVANOV, Talanta, 14 (1967) 171.
T. OZAKI, Anal. Lett., 15A (1982) 581.
L. SOMMER, T. SEPEL, V. M. IVANOV, Talanta, 15 (1968) 949.
K. KASIURA, M. SZCZYGIESKA, Chemia Analyt., 16 (1971) 671.
K. KASIURA, J. M. MINCZEWSKI, Nucleonika, 11 (1966) 299.
S. I. GUSEV, V. A. AGILOV, L. M. SHUROVA, Zh. Analit. Khim., 30 (1975) 540.
C. D. SHARMA, M. C. ESHWAR, J. Radioanal. Nucl. Chem., 91 (1985) 323.
G. V. RATHAIAH, M. C. EESHWAR, Bull. Chem. Soc. Japan, 58 (1985) 2447.
G. V. RATHAIAH, M. C. ESHWAR, Indian J. Techn., 23 (1985) 157.
A. I. VOGEL, A. Text-book of Quantitative Inorganic Analysis, Longman, London, 1968, p. 540.
B. V. KADAM, B. MAITI, R. M. SATHE, Analyst, 106 (1981) 724.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rathaiah, G.V., Charyulu, J.K. & Eshwar, M.C. Spectrophotometric determination of uranium(VI) with 1-(2′-thiazolylazo)-2-naphthol in the presence of Trition X-100. Journal of Radioanalytical and Nuclear Chemistry, Articles 99, 337–344 (1986). https://doi.org/10.1007/BF02037594
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02037594