Abstract
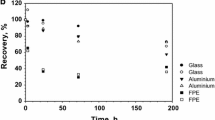
Solutions of Pb2+ at ppb to ppm levels were tagged with210Pb and then counted from time to time over a period of one year to measure the extent of container adsorption. All count rates for samples which were acidified with HNO3 were at least 90% of their initial values, even after one year of storage. Amber glass containers proved to be superior to polypropylene for 0.1 and 1.0 ppm Pb2+ solutions; but below 0.1 ppm, polypropylene was superior. For non-acidified samples, the count rate dropped rapidly within the first two months, and then declined more gradually thereafter, with polypropylene generally superior.
Similar content being viewed by others
References
D. E. ROBERTSON, Anal. Chim. Acta, 42 (1968) 533.
R. MASSEE, F. J. M. J. MASSEN, Anal. Chim. Acta, 127 (1981) 181.
A. W. STRUEMPLER, Anal. Chem., 45 (1973) 2251.
G. E. BATLEY, D. GARDNER, Water Res., 11 (1977) 745.
K. S. SUBRAMANIAN et al., Anal. Chem., 50 (1978) 444.
G. E. BATLEY, T. M. FLORENCE, Marine Chem., 4 (1976) 347.
L. M. PETRIE, R. W. BAIER, Anal. Chem., 50 (1978) 351.
R. A. PACER, J. Radioanal. Chem., 77 (1983) 19.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pacer, R.A., Weber, T.J. The role of210Pb in evaluating container adsorption of Pb(II) from dilute aqueous solutions. Journal of Radioanalytical and Nuclear Chemistry, Articles 129, 181–190 (1989). https://doi.org/10.1007/BF02037581
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02037581