Abstract
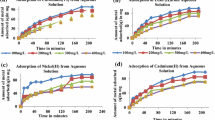
The adsorption of metal ions such as Ce3+, Sm3+, Eu3+ and Gd3+ ions on activated charcoal has been studied as a function of pH. The adsorption mechanism of these ions is discussed in terms of hydrolyzed species formed in aqueous solution at different pH.
Similar content being viewed by others
References
H. C. PARREIRA, J. H. SCHULMAN, Advan. Chem. Ser., 33 (1961) 160.
R. H. OTTEWILL, J. N. SHAW, Kolloid-Z. Z. Polym., 218 (1967) 34.
P. BENES, J. PAULENOVA, Kolloid-Z. Z. Polym., 251 (1973) 766.
P. BENES, J. SINGER, Collect. Czech. Chem. Commun., 33 (1968) 1508.
P. BENES, J. KUCERA, Collect. Czech. Chem. Commun., 37 (1972) 523.
R. C. VICKERY, Nature, 158 (1946) 623.
I. S. KIRIN, V. K. VESELOV, A. F. IVANCHENKO, Radiokhimiya, 17 (1975) 488.
M. AFZAL, J. HANIF, I. HANIF, R. QADEER, M. SALEEM, J. Radioanal Nucl. Chem., 139 (1990) 203.
J. KRAGTEN, Atlas of Metal-Ligand Equilibria in Aqueous Solutions, Horwood, Chichester, 1978.
F. ICHIKAWA, T. SATO, Radiochim. Acta 12 (1969) 89.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Saleem, M., Qadeer, R., Hanif, J. et al. Effect of pH on the adsorption of Ce3+, Sm3+, Eu3+ and Gd3+ ions on activated charcoal. Journal of Radioanalytical and Nuclear Chemistry, Articles 182, 415–419 (1994). https://doi.org/10.1007/BF02037518
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02037518