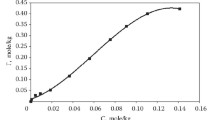
Abstract
The ion-exchange process on columns filled with granulated zeolites is determined by several physico-chemical parameters. The influence of these parameters (zeolite type, concentration of exchangeable ions in solution, temperature, flow rate, etc.) on the kinetics of ion-exchange process was studied by measuring the Sr2+ ion concentration in solution before and after passing through a column filled with various granulated zeolites (zeolite 13X, zeolite A and synthetic mordenite). Using the experimental technique of radioactive labeling by89Sr, the distribution of Sr2+ ions in column fillings were also determined. From the results obtained, the optimal conditions for the most efficient removal of strontium ions from solutions using granulated zeolites can be defined.
Similar content being viewed by others
References
H. K. MANAKTALA, Nuclear Waste Management Technical Support, in: Development of Nuclear Waste Form Criteria for the NCR, Vol. 3, Waste Inventory Review, Brookhaven National Laboratory, Upton, 1982.
J. O. LILJEZIN, G. PERSSON, I. HAGSTRÖM, I. SVANTESSON, Sci. Basis Nucl. Waste Managem., 2 (1980) 885.
M. KUBOTA, I. YAMAGUCHI, K. OKADA, Y. MORITA, K. NAKANO, H. NAKAMURA, Proc. Symp. Mat. Res. Soc., 26 (1984) 551.
H. MIMURA, T. KANNO, J. Nucl. Sci. Technol., 22 (1985) 284.
IAEA, Technical Report Series No. 222, Conditioning of Low and Intermediate Level Radioactive Waste, International Atomic Energy Agency, Vienna, 1983.
K. W. CARLEY-MACANLY, Proc. Int. Congr. Radioactive Waste Management, Vol. 2, International Atomic Energy Agency, Vienna, 1984, p. 15.
S. FORBERG, T. WESTERMARK, L. FÄLTH, Basic Nucl. Waste Managem., 3 (1981) 227.
J. BRONIĆ, B. SUBOTIĆ, J. Radioanal. Nucl. Chem., 100 (1986) 91.
B. SUBOTIĆ, J. BRONIĆ, J. Radioanal. Nucl. Chem., 102 (1986) 465.
J. BRONIĆ, B. SUBOTIĆ, J. Radioanal. Nucl. Chem., 152 (1992) 359.
T. KANNO, H. HASIMOTO, Bull. Res. Inst. Min. Dress. Met., 32 (1976) 14.
T. KANNO, H. HASIMOTO, T. OTANI Bull. Res. Inst. Min. Dress. Met., 34 (1978) 53.
H. MIMURA, T. KANNO, Sci. Rep. RITU, A30 (1981) 125.
M. J. SEMMENS, in Zeo-Agriculture, W. G. POND, F.A. MUMPTON (Eds), Westview Press, Boulder Colorado, 1894, p. 45.
H. MIMURA, M. FUKUMORI, T. OTANI, T. KANNO, Bull. Res. Inst. Min. Dress. Met., 35 (1979) 19.
H. MIMURA, T. KIMURA, T. KANNO, Bull. Res. Inst. Min. Dress. Met., 37 (1981) 145.
A. NASTRO, C. COLLELA, Inorg. Chim. Ital., 19 (1983) 41.
J. BRONIC, M. Sc. Thesis, University of Zagreb, 1989.
M. J. SCHWUGER, H. G. SMOLKA, Tenside-Detergents, 13 (1976) 305.
K. R. FRANKLIN, R. P. TOWNSEND, J. Chem. Soc. Faraday Trans., 1 81 (1985) 1071.
H. MIMURA, T. KANNO, Bull. Res. Inst. Min. Dress. Met., 34 (1978) 58.
H. MIMURA, I. ISEKI, K. KUDA, T. KANNO, Bull. Res. Inst. Min. Dress. Met., 40 (1984) 38.
C. CAPELLOS, B. H. BIELSKI, Kinetic Systems, Wiley-Interscience, 1972, p. 113.
H. MIMURA, M. FUKUMORI, T. OTANI, T. KANNO, Bull. Res. Inst. Min. Dress. Met., 35 (1979) 19.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bronić, J., Subotić, B. Removal of strontium ions from solutions using granulated zeolites. Journal of Radioanalytical and Nuclear Chemistry, Articles 162, 339–350 (1992). https://doi.org/10.1007/BF02035394
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02035394