Abstract
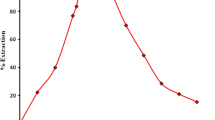
The use of Amberlite-LA-2, a high molecular weight secondary amine, for the radiochemical solvent extraction of Ag(I) present as anionic thiosulfato complex at pH 3 is described. The effects of different parameters on the extraction and preconcentration have been studied in detail. The method was successfully applied in the determination of silver in several synthetic mixutres and also in a few medicinal samples.
Similar content being viewed by others
References
H. GREEN, Talanta, 20 (1973) 139.
T. SATO, J. Inorg. Nucl. Chem., 28 (1966) 1461.
A. CHATTERJEE, S. BASU, Indian J. Chem., 25A (1990) 1117.
A. CHATTERJEE, S. BASU, J. Indian Chem. Soc., 67 (1990) 895.
T. GROCHEWALD, Anal. Chem., 41 (1969) 1012.
D. C. MEDIGAN, Aust. Miner Develop. Lab. Bull., 6 (1968) 61.
A. ALICE, Michrochim. Acta, 9 (1968) 1081.
F. SALINAS, B. NERADO, J. J. ESPINOSA, A. MANSILLA, T. G. ROMA, Quim. Anal. (Barcelona), 6 (1987) No. 2, 232.
E. TAKASHI, K. KOJIMA, Japan Assoc. Proc. Symp. Solvent Extr. 133 1988, p. 133.
C. FISCHER, H. WAGNER, N. V. V. BAGREE, J. Inorg. Nucl. Chem., 42 (1980) 891.
A. CHATTERJEE, S. BASU, J. Radioanal. Nucl. Chem., 150 (1990) 177.
K. KODAMA, Quantitative Inorganic Analysis, Elsevier, Amsterdam, 1963, p. 299.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chatterjee, A., Basu, S. Solvent extraction of silver(I) with amberlite-LA-2 using110mAg as tracer. Journal of Radioanalytical and Nuclear Chemistry, Articles 162, 259–265 (1992). https://doi.org/10.1007/BF02035386
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02035386