Summary
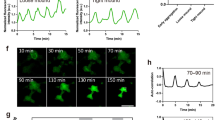
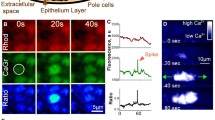
Both the natural metamorphic stimulus (an unidentified bacterial product) and an artificial trigger of metamorphosis (Cs+) cause large calcium transients in planula cells of the hydrozoanMitrocomella polydiademata. When these transients are inhibited with calcium channel blockers, metamorphosis is also inhibited. All cells of theMitrocomella planula contain a calcium-specific photoprotein. The cells where the calcium transients occur during natural- and Cs+-induced metamorphosis have been visualized in normal and entoderm free planulae that lack ganglion cells, using a compound microscope coupled to an image intensifier and video camera. During bacteria- and Cs+-induced metamorphosis, groups of contiguous cells, occupying from about 10% to the entire visible surface of the planula, simultaneously exhibit calcium transients. When the cells that initiate a transient comprise only part of the planula surface, the calcium transient frequently propagates and can eventually involve every cell on the visible planula surface. There is no special site on the planula surface where calcium transients are more apt to be initiated. There is no indication that propagation of a flash in one direction is more likely than in another. The velocity of propagation is virtually the same in all directions. The only feature of the spatial distribution of bacteria- and Cs+-induced calcium transients that appears to be necessary for the induction of metamorphosis is that at least one transient must involve all of the surface cells of the planula. The spatial behavior of calcium transients is the same in entoderm free planulae (lacking ganglion cells) as in normal planulae. The propagation of these calcium transients most probably occurs via epithelial conduction. This metamorphic step involving calcium transients is probably the intercellular communication system that informs the cells of the planula that metamorphosis will commence.
Metamorphosis inMitrocomella planulae can also be induced with phorbol esters. Calcium transients do not occur during phorbol ester-induced metamorphosis, indicating that they act at a different point in the metamorphic pathway. Calcium channel blockers do not inhibit phorbol ester-induced metamorphosis. Inhibitors of protein kinase-C, inhibit both phorbol ester-induced metamorphosis and Cs+- and bacteria-induced metamorphosis, but have no effect on the calcium transients induced by Cs+. This indicates that the calcium transient mediated step in the metamorphic pathway occurs prior to protein kinase-C activation. Calcium transients probably play a major role in activating protein kinase-C.
Similar content being viewed by others
References
Alkon DL, Rasmussen H (1988) A spatial-temporal model of cell activation. Science 239:998–1005
Anderson PAV (1980) Epithelial conduction: its properties and functions. Prog Neurobiol 15:161–203
Bassot J-M, Bilbaut A, Mackie GO, Passano LM, Pavans de Ceccatty M (1978) Bioluminescence and other responses spread by epithelial conduction in the siphonophoreHippopodius. Biol Bull 155:473–498
Berking S (1988) Ammonia, tetraethylammonium, barium, and amiloride induce metamorphosis in the marine hydroidHydractinia. Roux's Arch Dev Biol 197:1–9
Edwards NC, Thomas MB, Long BA, Amyotte SJ (1987) Catecholamines induce metamorphosis in the hydrozoanHalocordyle disticha but notHydractinia echinata. Roux's Arch Dev Biol 196:381–384
Freeman G (1981) The role of polarity in the development of the hydrozoan planula larva. Roux's Arch Dev Biol 190:168–184
Freeman G, Ridgway EB (1987) Endogenous photoproteins, calcium channels, and calcium transients during metamorphosis in hydrozoans. Roux's Arch Dev Biol 196:30–50
Freeman G, Ridgway EB (1988) The role of cAMP in oocyte maturation and the role of the germinal vesicle contents in mediating maturation and subsequent developmental events in hydrozoans. Roux's Arch Dev Biol 197:197–211
Hille B (1984) Ionic Channels of Excitable Membranes. Sinauer Assoc, Sunderland, Mass, pp 1–426
Horn F, Gschwendt M, Marks F (1985) Partial purification and characterization of the calcium-dependent and phospholipiddependent protein Kinase C from chick oviduct. Eur J Biochem 148:533–538
Kiley SG, Jaken S (1990) Activation of a protein kinase C leads to association with detergent-insoluble components in GH4G cells. Mol Endocrinal (in press)
Kolberg K, Martin V (1988) Morphological, cytochemical and neuropharmacological evidence for the presence of catecholamines in hydrozoan planulae. Development 103:249–258
Leitz T, Müller WA (1987) Evidence for the involvement of PI-signaling and diacylglycerol second messengers in the initiation of metamorphosis in the hydroidHydractinia echinata. Fleming. Dev Biol 121:82–89
Mackie GO, Anderson PAV, Singla CL (1984) Apparent absence of gap junctions in two classes of cnidaria. Biol Bull 167:120–123
Martin VJ (1988a) Development of nerve cells in hydrozoan planulae: I. Differentiation of ganglion cells. Biol Bull 174:319–329
Martin VJ (1988b) Development of nerve cells in hydrozoan planulae: II. Examination of sensory cell differentiation using electron microscopy and immunocytochemistry. Biol Bull 175:65–78
Martin VJ, Thomas MB (1981) Elimination of the interstitial cells in the planula of the larva of the marine hydrozoanPennaria tiarella. J Exp Zool 217:313–323
Martin V, Chia F-S, Koss R (1983) A fine structural study of metamorphosis of the hydrozoanMitrocomella polydiademata. J Morphol 176:261–287
Martin VJ, Archer WE (1986) Migration of interstitial cells and their derivatives in a hydrozoan planula. Dev Biol 116:486–496
May WS, Sahyoun N, Wolf M, Cutrecasas P (1985) Role of intracellular calcium mobilization in the regulation of protein kinase-C-mediated membrane processes. Nature 317:549–551
Müller WA (1973) Metamorphose-Induction bei Planulalarven I. Der Bakterielle Induktor. Roux' Arch Dev Biol 173:107–121
Müller WA (1985) Tumor-promoting phorbol esters induce metamorphosis and multiple head formation in the hydroidHydractinia. Differentiation 29:216–222
Müller WH, Buchal G (1973) Metamorphose-Induktion bei Planulalarven II. Induktion durch monovalente Kationen: Die Bedeutung des Gibbs-Donnan-Verhältnisses und der Na+/K+ ATPase. Roux' Arch Dev Biol 173:122–135
Schwoerer-Böhning B, Kroiher M, Müller WA (1990) Signal transmission and covert prepattern in the metamorphosis ofHydractinia echinata (Hydrozoa). Roux's Arch Dev Biol 198:245–251
Spencer A (1974) Non-nervous conduction in invertebrates and embryos. Am Zool 14:917–929
Thomas MB, Freeman G, Martin VJ (1987) The embryonic origin of neurosensory cells and the role of nerve cells in metamorphosis inPhialidium gregarium (Cnidaria, Hydrozoa). Int J Invert Reprod Dev 11:265–287
Weis V, Keene D, Buss L (1985) Biology of the hydractiniid hydroids 4. Ultrastructure of the planula ofHydractinia echinata. Biol Bull 168:403–418
Wolf M, LeVine H, May WS, Cuatrecasas P, Sahyoun N (1985) A model for intracellular translocation of protein kinase-C involving synergism between Ca2+ and phorbol esters. Nature 317:546–559
Yool AJ, Gray SM, Hadfield MG, Jensen RA, Markell DA, Morse DE (1986) Excess potassium induces metamorphosis in four marine invertebrate species. Biol Bull 170:255–265
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Freeman, G., Ridgway, E.B. Cellular and intracellular pathways mediating the metamorphic stimulus in hydrozoan planulae. Roux's Arch Dev Biol 199, 63–79 (1990). https://doi.org/10.1007/BF02029553
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02029553