Abstract
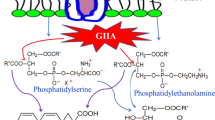
The three types (groups I, II and III) of stable extracellular 14 kDa phospholipase A2 enzymes differ in their primary amino acid sequences and their properties. It may thus be possible to design low-molecular weight inhibitors targeted to the secretory form of mammalian PLA2. this enzyme has been implicated in inflammatory disorders. We have studied the inhibition of four distinct PLA2 enzymes by a range of NSAIDs, using3H-oleate release from prelabelled membranes ofE. coli for assay. The enzymes used were cobra venom PLA2 (Naja naja, a group I enzyme), bee venom PLA2 (Apis mellifera, group III), recombinant human synovial PLA2 (group II) and rat peritoneal PLA2 (group II). Under the conditions of the3H-oleateE. coli assay, 1 mM concentrations of aspirin, sodium salicylate, paracetamol (acetaminophen), oxphenbutazone, ibuprofen, flurbiprofen and nabumetone failed to inhibit significantly any of the four enzymes. However, indomethacin inhibited all four enzymes, although effects were greatest on the two group II enzymes (rat peritoneal and human synovial PLA2). Approximate IC50 values were 28 and 35 μM, respectively. Inhibition by indomethacin was not time dependent and was greater at micromolar rather than millimolar levels of calcium. We conclude that indomethacin but not the other tested classes of NSAID inhibits the group II PLA2 enzyme in a selective manner and suggest that this may be relevant both to its clinical spectrum and to the design of novel pharmaceutical leads.
Similar content being viewed by others
References
H. M. Verheij, H. J. Slotboom and G. H. de Hass,Structure and function of phospholipase A 2. Rev. Physiol. Biochem. Pharmacol.91, 91–203 (1981).
E. A. Dennis,Phospholipases. InThe Enzymes, vol. 16. (Ed. P. D. Boyer) pp. 307–353, Academic Press, New York 1983.
P. Vadas and W. Pruzanski,Biology of disease: The role of secretory phospholipase A 2 in the pathobiology of disease. Lab. Invest.55, 391–404 (1986).
F. F. Davidson and E. A. Dennis,Evolutionary relationships and implications for the regulation of phospholipase A 2 from snake venom to human secreted forms. J. Mol. Evol.31, 228–238 (1990).
H. van den Bosch,Intracellular phospholipases A. Biochim. Biophys. Acta604, 191–246 (1980).
L. Kaplan, J. Weiss and P. Elsbach,Low concentrations of indomethacin inhibit phospholipase A 2 of rabbit polymorphonuclear leukocytes. Proc. Natl. Acad. Sci. USA75, 2955–2958 (1978).
R. L. Jesse and R. C. Franson,Modulation of purified phospholipase A 2 activity from human platelets by calcium and indomethacin. Biochim. Biophys. Acta575, 467–470 (1979).
R. C. Franson, D. Eisen, R. Jesse and C. Lanni,Inhibition of highly purified mammalian phospholipases A 2 by non-steroidal anti-inflammatory agents. Modulation by calcium ions. Biochem. J.186, 633–636 (1980).
L. Kaplan-Harris and P. Elsbach,The antiinflammatory activity of analogs of indomethacin correlates with their inhibitory effects on phospholipase A 2 of rabbit polymorphonuclear leukocytes. Biochim. Biophys. Acta618, 318–326 (1980).
P. Patriarca, S. Beckerdite and P. Elsbach,Phospholipases and phospholipid turnover in Escherichia colispheroplasts. Biochim. Biophys. Acta260, 593–600 (1972).
L. A. Marshall, J. Bauer, M. L. Sung and J. Y. Chang,Evaluation of antirheumatic drugs for their effect in vitro on purified human synovial fluid phospholipase A 2. J. Rheumatol.18, 59–65 (1991).
B. S. Vishwanath, A. A. Fauzy and R. C. Franson,Edemainducing activity of phospholipase A 2 purified from human synovial fluid and inhibition by aristolochic acid. Inflammation12, 549–561 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lobo, I.B., Hoult, J.R.S. Groups I, II and III extracellular phospholipases A2: Selective inhibition of group II enzymes by indomethacin but not other NSAIDs. Agents and Actions 41, 111–113 (1994). https://doi.org/10.1007/BF01986409
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01986409