Abstract
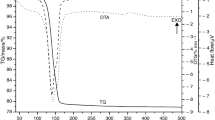
The dehydroxylation of gibbsite into boehmite was investigated by means of DSC analysis under non-isothermal conditions in the temperature range 453–673 K at heating rates from 2.5 to 20.0 K min−1. Mathematical analysis of the experimental DSC curves revealed the mechanism and kinetics of the gibbsite dehydroxylation process. The kinetic curvesα=f(t) andα=f(T) are sigmoidal in shape; their inflection points and the νm point of the curvesν=f(T) andν=f(T) are interrelated and are defined by the concept of a stationary point. The activation energy for the first stage of gibbsite dehydroxylation in the temperature range 453–673 K is 132.92±8.33–142.26±8.33 kJ mol−1.
Similar content being viewed by others
References
V. Šatava, Thcrmochim. Acta, 2 (1971) 423.
R. Vračar, M. Tecilazić-Stevanović, D. Sinadinović and T. Janačković, 4th International Congress ICSOBA, Athens, 1978, Vol. 2, 937/950.
D. Blečić, Ž. Živković and M. Martinović, Thermochim. Acta, 60 (1983) 61.
D. Blečić and Ž. Živković, 3. sav. probl. proizv., prer. i potr. boks., glinice i alum. Jug., Šibenik 1984, Zb. rad., 243/252.
J. Perić, R. Krstulović, T. Ferić, M. Vučak, Travaux ICSOBA, 20–21 (1991) 35.
DSC-4 Standard Program Manual (Perkin-Elmer Part No. 0993-9823).
M. Antić and N. Colović, Kinetika heterogenih hemijskih reakcija, Institut zaštite na radu. Niš, 1083, 269/278.
J. Perić, R. Krstulović, T. Ferić and M. Vučak, Thermochim. Acta, 207 (1992) 245.
L. Candela and D. D. Perlmutter, Ind. Eng. Chem. Res., 31 (1992) 694.