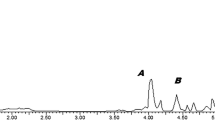
Summary
The ethereal extract of the mucous secretion from the opisthobranch molluscOxynoe olivacea was examined and found to contain two novel ichthyotoxic compounds, named oxytoxin 1 and 2 (1, 2). The structures of1 and2 are closely related to the metabolites previously isolated from the algaCaulerpa prolifera. The activity of the most stable compound was studied in order to investigate the possibility, of a further biological role for these metabolites, which represent an uncommon example of bioactive molecules produced in vivo from a dietary precursor.
Similar content being viewed by others
References
Lewin, R. A., Pacific Sci.24 (1970) 356.
Warmke, G. L., and Almodovar, L. R., Bull. mar. Sci.22 (1972) 67.
Stamm, R. A., Revue suisse Zool.75 (1968) 661.
Doty, M. S., and Aguilar-Santos, G., Pacific Sci.24 (1970) 351.
Marin, A., and Ros, J. D., Iberus8 (1988) 25; Schmekel, L., and Portmann, A., Opistobranchia des Mittelmeeres (Nudibranchia und Sacoglossa) Springer Verlag, Berlin 1982.
Amico, V., Oriente, G., Piattelli, M., Tringali, C., Fattorusso, E., Magno, S., and Mayol, L., Tetrahedron Lett.38 (1978) 3593.
De Napoli, L., Fattorusso, E., Magno, S., and Mayol, L., Experientia37 (1981) 1132.
De Napoli, L., Magno, S., Mayol, L. and Novellino, E., Experientia39 (1983) 141.
Gunthorpe L., and Cameron, A. M., Mar. Biol.94 (1987) 39. The toxicity ranking was defined according to Coll, J. C., La Barre, S., Sammarco, P. W., Williams, W. T., and Bakus, G., Mar. Ecol. Prog. Ser.8 (1982) 271.
Ireland, C., and Faulkner, D. J., Bioorg. Chem.7 (1978) 125.
Paul, V. J., and Fenical, W., Tetrahedron40 (1984) 2913.
Cimino, G., Sodano, G., and Spinella, A., J. nat. Prod.51 (1988) 1010.
Cimino, G., De Rosa, S., De Stefano, S., and Sodano, G., Comp. Biochem. Physiol.73B (1982) 471.
Meyer, B. N., Ferrigni, N. R., Putnam, J. E., Jacobsen, L. B., Nichols, D. E., and McLaughlin, J. L., Planta med.45 (1982) 31.
De Petrocellis, L., Maharajan, V., De Petrocellis, B., and Minei, R., J. Embryol. exp. Morph.93 (1986) 105. Burnett, A. L., Am. Nat.100 (1966) 165.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cimino, G., Crispino, A., Di Marzo, V. et al. Oxytoxins, bioactive molecules produced by the marine opisthobranch molluscOxynoe olivacea from a diet-derived precursor. Experientia 46, 767–770 (1990). https://doi.org/10.1007/BF01939963
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01939963