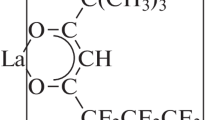
Abstract
The multi-step dehydration and decomposition of trivalent lanthanum and lanthanide heptanediate polyhydrates were investigated by means of thermal analysis completed with infrared study. Further more, X-ray diffraction data for investigated heptanediate complexes of general stoichiometry Ln2(C7H10O4)3.nH2O (wheren=16 in the case of La, Ce, Pr, Nd and Sm pimelates,n=8 for Eu, Gd, Tb, Dy, Er and Tm pimelates,n=12 for Ho, Yb and Lu pimelates) were also reported.
Zusammenfassung
Mittels TG, DTG, DTA wurde in Verbindung mit IR-Methoden der mehrstufige Dehydratations- und der Zersetzungsvorgang der Polyhydrate der PimelinsÄuresalze von dreiwertigem Lanthan und dreiwertigen Lanthanoiden untersucht. Röntgendiffraktionsdaten der untersuchten Heptandiat-Komplexe mit der allgemeinen Formel Ln2(C7H10O4)3 nH2O (mitn=16 für Ln=La, Ce, Pr, Nd und Sm,n=8 für Ln=Eu, Gd, Tb, Dy, Er und Tm sowien=12 für Ln=Ho, Yb und Lu) werden ebenfalls gegeben.
Similar content being viewed by others
References
Beilsteins Handbuch der Organischen Chemie. B.2, Berlin 1920, p. 670; I. ErgÄnzungswerk, Ann Arbor-Michigan 1942, p. 670; II. Erg., Ann Arbor-Michigan 1953, p. 586; III. Erg., Berlin-Götingen-Heidelberg 1961, p. 1740; IV. Erg, Berlin-Heidelberg-New York 1976, p. 2003.
P. E. Verkade, Z. physiol, Chem., 237 (1935) 188.
A. Müller, J. Biol. Chem., 119 (1937) 1.
T. Chuman and M. Noguchi, Agric. Biol. Chem., 41 (1977) 1021; Chem. Abstr., 87 (1977) 84419 q.
R. Chauvin, J. P. Lafarge, J. P. Saligot, Comp. Rend. Acad. Sci., Ser. 3 (1984) 299.
N. Emmrich and N. Nebe, Z. physiol. Chem., 266 (1940) 179.
J. P. Jordan, M. Bounias and R. Chauvin, Comp. Rend. Seances Soc. Ses Fil., 181 (1987) 314; Chem. Abstr., 107 (1987) 195360 a.
C. Agouridas, J. M. Girodeau, R. Pineau and F. Le Goffie, French Pat. 2.566,410, 20 June 1984; Chem. Abstr., 105 (1986) 173044 g.
S. Iida, K. Kita, H. Otsuki, K. Hasegava, N. Asai and T. Eguchi; Japan Kokai Tokkyo Koho, Jap. Pat. 61.143,317, 1 July 1986; Chem. Abstr., 105 (1986) 139451 c.
M. Kimura, J. Suzuki, K. Waki, H. Matsuura, T. Fuva, R. Kasai and O. Tanaka; Japan Kokai Tokyo Koho, Jap. Pat. 61.171,417, 2 August 1986; Chem. Abstr., 105 (1986) 183975 a.
S. V. Grishchenko, A. M. Gazieva and O.V. Podlepyan, Nefteperab. Neftekhim. (Moscow), 6 (1980) 41; Chem. Abstr., 93 (1980) 244769 u.
D. J. Ryabchikov and E. A. Terentieva, fewest. Akad. Nauk SSSR, (1949) 44.
W. Brzyska and W. Hubicki, Ann. Univ. M. Curie-Sklodowska, Lublin, Sectio AA, 23 (1968) 83; Chem. Abstr., 71 (1969) 116998 b.
B. S. Azikov and W. W. Serebrennikov, Trudy Tomsk. Gos. Univ. Seria Khim., 185 (1965) 72, 111, 118; 192 (1968) 71, 74; Chem. Abstr., 66 (1967) 51814 p, 43328 t, 43327 s; 73 (1970) 72665 a, 74 (1971) 111400 c.
A. I. Turova and W. W. Serebrennikov, Trudy Tomsk. Gos. Univ. Seria Khim., 264 (1975) 74.
M. Dabkowska and M. Sikorska, Folia Sci. Soc. Lublinensis, Mat. Fiz. Chem., 29 (1988) in press. Preliminary note.
A. J. Vogel, J. Chem. Soc., (1929) 722.
K. Nakamoto, Infrared Spectra of Inorganic and Coordination Compounds J. Wiley, New York-London 1963.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dabkowska, M. Thermal behaviour of hydrated lanthanide complexes with heptanedioic acid. Journal of Thermal Analysis 36, 1319–1330 (1990). https://doi.org/10.1007/BF01914055
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01914055