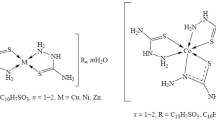
Abstract
Several zinc(II) complexes of tridentate dibasic salicylidene amino acids have been prepared and characterized. All of them possess dimeric pseudo-octahedral structures, which were established on the basis of thermogravimetric analysis and superimposable infrared spectra of these complexes with those of manganese(II), cobalt(II), nickel(II), and magnesium(II) complexes.
Zusammenfassung
Einige Zink(II)-komplexe von dreizähligen, dibasischen Salizylidenaminosäuren wurden dargestellt und beschrieben. Alle diese Komplexe besitzen eine pseudooktaedrische Dimerstruktur. Dies wurde mittels thermogravimetrischer Analyse und auf Grund der deckungsgleichen IR-Spektren dieser Komplexe mit denen von Mangan(II)-, Kobalt(II)-, Nickel(II)- und Magnesium(II)-Komplexen festgestellt.
Резюме
Получены и охарактер изованы несколько комплексов цинка с тр идентатными двухосновными салиц илиденаминокислота ми. Все комплексы обладают д имерной псевдооктаэдрическ ой структурой, устано вленной на основе термогравиме трического анализа и сопоставлением ИК сп ектров этих комплекс ов с таковыми для двухвал ентных марганца, кобальта, никеля и маг ния.
Similar content being viewed by others
References
R. L. Dutta and R. K. Ray, J. Inorg. Nucl. Chem., 39 (1977) 1848.
R. Pani and B. Behera, Indian J. Chem., 12 (1974) 215.
L. J. Theriot, G. O. Carlisle and H. J. Hue, J. Inorg. Nucl. Chem., 31 (1969) 2891.
R. L. Dutta and R. K. Ray, J. Indian Chem. Soc., 54 (1977) 1096; 60 (1983) 185.
R. L. Dutta and R. K. Ray, J. Inorg. Nucl. Chem., 41 (1979) 755.
R. H. Holm, G. W. Everett, Jr. and A. Chakraborty, Progress in inorganic chemistry, F. A. Cotton, Ed., Interscience Publishers, New York, 1966; Vol. 7, p. 83.
K. Nakamoto, Infrared spectra of inorganic and coordination compounds, John Wiley, New York, 1966.
L. J. Boucher and C. G. Coe, Inorg. Chem., 14 (1975) 1289.
N. S. Biradar and V. K. Kulkarni, J. Inorg. Nucl. Chem., 33 (1971) 2451.
S. G. Gruber, C. N. Harris and E. Sinn, J. Inorg. Nucl. Chem., 30 (1968) 1805.
T. Tokii, Y. Muto, M. Kato, K. Imal and H. Jonassen, J. Inorg. Nucl. Chem., 34 (1972) 3377.
H. H. Freedman, J. Am. Chem. Soc., 83 (1961) 2900.
B. Kim, C. Miyake and S. Imoto, J. Inorg. Nucl. Chem., 37 (1975) 963.
C. Duval, Inorganic thermogravimetric analysis, 2nd ed., Elsevier Publishing Co., New York, 1963; pp. 312–350.
Author information
Authors and Affiliations
Additional information
The senior author (RKR) expresses his deep gratitude to Prof. R. L. Dutta, Department of Chemistry, Burdwan University, India, for valuable suggestions and discussions during the course of the work. He is also grateful to Dr. R. N. Banerjee of Jadavpur University and Dr. P. Banerjee of the Indian Association for the Cultivation of Science, Calcutta for laboratory and other facilities.
Rights and permissions
About this article
Cite this article
Ray, R.K., Kauffman, G.B. Zinc(II) complexes of salicylidene amino acids. Journal of Thermal Analysis 35, 1603–1609 (1989). https://doi.org/10.1007/BF01912935
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01912935