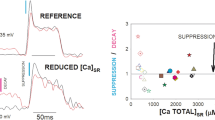
Summary
An increase in extracellular potassium concentration from 4 to 16 mmol/l caused a decrease in membrane potential from −92 to −59 mV and selectively diminished the earlier of two contraction components of guinea-pig papillary muscles at 0.2 Hz stimulation frequency in the presence of noradrenaline. The influence on the early contraction component had a threshold of 8 mmol/l K+, corresponding to a membrane potential of −77 mV. However, test contractions elicited 800 ms after the 5 s stimulation interval exhibited an unimpaired early component. Since the activator calcium responsible for the early contraction component is derived, in mammalian ventricular muscle, from the junctional sarcoplasmic reticulum (20), it is assumed that the release site of the reticulum was filled with calcium shortly (800 ms) after a regular contraction, and lost its calcium at 16 mmol/l extracellular K+ during the 5 s stimulation interval. The potassium-induced depolarization determined the rate of calcium leakage during rest from the intracellular store. The depolarization-induced decline of the early contraction component was equally well antagonized by Mg2+ or Ca2+ without influencing the measured transmembrane potential. Both divalent cations shifted the relation between potassium concentration or membrane potential and the strength of the early contraction component to less negative membrane potentials. In order to reduce the early contraction component by 25% in the presence of 9.6 instead of 1.2 mmol/l Mg2+, the potassium concentration had to be increased from 9.6 to 22.0 mmol/l, with a respective decrease in resting membrane potential from −72.6 to −51.1 mV. The antagonistic effect of both divalent cations is thought to result from the neutralization of negative charges outside the sarcolemma with a respective decrease in the outside surface potential.
Similar content being viewed by others
References
Allen DG, Jewell BR, Wood EH (1976) Studies of the contractility of mammalian myocardium at low rates of stimulation. J. Physiol 254:1–17
Bianchi CP, Narayan S, Lakshminarayanaiah N (1975) Mobilization of muscle calcium and oxygen uptake in skeletal muscle. In: Carafoli E (ed) Calcium Transport in Contraction and Secretion. North-Holland Publishing Company Amsterdam pp 503–515
Chandler WK, Hodgkin AL, Meves H (1965) The effect of changing the internal solution on sodium inactivation and related phenomena in giant axons. J Physiol 180:821–836
Eisenberg BR, Eisenberg RS (1982) The T-SR junction in contracting single skeletal muscle fibers. J Gen Physiol 79:1–19
Endo M, Kitazawa T (1978) E-C coupling studies on skinned cardiac fibers. In: Morad M (ed) Biophysical Aspects of Cardiac Muscle. Academic Press, New York, pp 307–327
Forbes MS, Sperelakis N (1982) Bridging junctional processes in couplings of skeletal, cardiac, and smooth muscle. Muscle and Nerve 5:674–681
Franzini-Armstrong C (1970) Studies of the triad. I. Structure of the junction in frog twitch fibers. J Cell Biol 47:488–499
Franzini-Armstrong C (1971) Studies of the triad. II. Penetration of tracers into the junctional gap. J Cell Biol 49:196–203
Hegnauer AH, Fenn WO, Cobb DM (1934) The cause of the rise in oxygen consumption of frog muscles in excess of potassium. J Cell Comp Physiol 4:505–526
Hille B, Woodhull AM, Shapiro BI (1975) Negative surface charge near sodium channels of nerve: divalent ions, monovalent ions, and pH. Phil Trans R Soc Lond B 270:301–318
Honerjäger P, Loibl E, Steidl I, Schönsteiner G, Ulm K (1986) Negative inotropic effects of tetrodotoxin and seven class 1 antiarrhythmic drugs in relation to sodium channel blockade. Naunyn-Schmiedeberg's Arch Pharmacol 332:184–195
Huxley AF (1971) The activation of striated muscle and its mechanical response. Proc R Soc Lond B 178:1–27
Kitazawa T (1984) Effect of extracellular calcium on contractile activation in guinea-pig ventricular muscle. J Physiol 355:635–659
Lewartowski B, Pytkowski B, Janczewski A (1984) Calcium fraction correlating with contractile force of ventricular muscle of guinea-pig heart. Pflügers Arch 401:198–203
Manzini S, Parlani M, Astolfi M, Maggi CA, Meli A (1986) Positive inotropic effect of Mg2+ in K+-depolarized isoprenaline-reactivated guinea-pig ventricular strip. Gen Pharmac 17:157–165
Mascher D, Cruz A (1980) Electrical and mechanical responses of the guinea-pig ventricular muscle in the presence of histamine. Proc Intern Union Physiol Sci 14:570
Mathias RT, Levis RA, Eisenberg RS (1980) Electrical models of exciation-contraction coupling and charge movement in skeletal muscle. J Gen Physiol 76:1–31
Novotny I, Vyskocil F (1966) Possible role of Ca ions in the resting metabolism of frog sartorius muscle during potassium depolarization. J Cell Physiol 67:159–168
Reiter M (1981) The use of the isolated papillary muscle for the evaluation of positive inotropic effects of cardioactive steroids. In: Greeff K (ed) The Handbook of Experimental Pharmacology, Vol 56/1 Springer, Berlin Heidelberg New York, pp 153–159
Reiter M, Vierling W, Seibel K (1984) Excitation-contraction coupling in rested-state contractions of guinea-pig ventricular myocardium. Naunyn-Schmiedeberg's Arch Pharmacol 325:159–169
Reiter M, Vierling W, Seibel K (1984) Where is the origin of the activator calcium in cardiac ventricular contraction? Basic Res Cardiol 79:1–8
Schneider MF, Chandler WK (1973) Voltage dependent charge movement in skeletal muscle: a possible step in excitation-contraction coupling. Nature 242:244–246
Seibel K, Reiter M (1978) Potassium-induced loss of the early component of myocardial contraction in presence of noradrenaline. Naunyn-Schmiedeberg's Arch Pharmacol 302:R31
Seibel K, Karema E, Takeya K, Reiter M (1976) Two components of heart muscle contraction under the influence of noradrenaline. Naunyn-Schmiedeberg's Arch Pharmacol 294:R19
Seibel K, Karema E, Takeya K, Reiter M (1978) Effect of noradrenaline on an early and a late component of the myocardial contraction. Naunyn-Schmiedeberg's Arch Pharmacol 305:65–74
Snowdowne KW, Lee NKM (1980) Subcontracture concentrations of potassium and stretch cause an increase in the activity of intracellular calcium in the frog skeletal muscle. Fed Proc 39:1733
Solandt DY (1936) The effect of potassium on the excitability and resting metabolism of frog's muscle. J Physiol 86:162–170
Sommer JR, Johnson EA (1979) Ultrastructure of cardiac muscle. In: Berne RM, Sperelakis N, Geiger RS (eds) The Handbook of Physiology—The Cardiovascular System 1 American Physiological Society, Bethesda, Maryland, pp 113–186
Van der Kloot WG (1967) Potassium-stimulated respiration and intracellular calcium release in frog skeletal muscle. J Physiol 191:141–165
Vierling W (1985) Magnesium abolishes potassium-, but not ryanodine-, induced suppression of contraction in the guinea-pig ventricular muscle. Naunyn-Schmiedeberg's Arch Pharmacol 330:R43
Vierling W, Seibel K, Reiter M (1986) Potential-dependent calcium release in cardiac ventricular muscle. Naunyn-Schmiedeberg's Arch Pharmacol 334:R31
Weidmann S (1955) The effect of the cardiac membrane potential on the rapid availability of the sodium-carrying system. J Physiol 127:213–224
Wendt-Gallitelli MF, Jacob R, Wolburg H (1982) Intracellular membranes as boundaries for ionic distribution. In situ elemental distribution in guinea pig heart muscle in different defined electromechanical coupling states. Z Naturforsch 37c: 712–720
Wiggins JR (1981) Inotropic actions of isoproterenol in cat ventricular muscle. Effects of extracellular potassium. Circ Res 49:718–725
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vierling, W., Seibel, K. & Reiter, M. Voltage-dependent calcium release in guinea-pig cardiac ventricular muscle as antagonized by magnesium and calcium. Basic Res Cardiol 82, 415–427 (1987). https://doi.org/10.1007/BF01907089
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01907089