Abstract
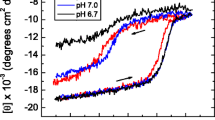
The refolding kinetics of guanidine-denatured disulfide-intact bovine pancreatic ribo-nuclease A (RNase A) and its proline-42-to-alanine mutant (Pro42Ala) have been studied by monitoring tyrosine burial and 2′-cytidine monophosphate (2′CMP) inhibitor binding. The folding rate for wild-type RNase A is faster in the presence of the inhibitor 2′CMP than in its absence, indicating that the transition-state structure in the rate-determining step is stabilized by 2′CMP. The folding rate monitored by 2′CMP binding to the major slow-folding species of Pro42Ala RNase A is faster than the folding rate monitored by tyrosine burial; however, the folding rate monitored by inhibitor binding to the minor slow-folding species is decreased significantly over the folding rate monitored by tyrosine burial, indicating that the major and minor slow-folding species of Pro42Ala fold to the native state with different transition-state conformations in the rate-determining step.
Similar content being viewed by others
References
Adler, M., and Scheraga, H. A. (1990).Biochemistry 29, 8211–8216.
Anderson, D. G., Hammes, G. G., and Walz, F. G., Jr. (1968).Biochemistry 7, 1637–1645.
Beaven, G. H., Holiday, E. R., and Johnson, E. A. (1955). InThe Nucleic Acids, Vol. 1 (Chargaff, E., and Davidson, J. N., eds.), Academic Press, New York, pp. 493–553.
Borkakoti, N., (1983).J. Biochem. 132, 89–94.
Borkakoti, N., Moss, D. S., and Palmer, R. A. (1983).Acta Cryst. B 38, 2210–2217.
Brandts, J. F., Halvorson, H. R., and Brennan, M. (1975).Biochemistry 14, 4953–4963.
Cook, K. H., Schmid, F. X., and Baldwin, R. L. (1979).Proc. Natl. Acad. Sci. USA 76, 6157–6161.
Crook, E. M., Mathias, A. P., and Rabin, B. R. (1960).Biochem. J. 74, 234–238.
Denton, J. B., Konishi, Y., and Scheraga, H. A. (1982).Biochemistry 21, 5155–5163.
Dyson, H. J., Rance, M., Houghten, R. A., Lerner, R. A., and Wright, P. E. (1988).J. Mol. Biol. 201, 161–200.
Garel, J.-R., and Baldwin, R. L. (1973).Proc. Natl. Acad. Sci. USA 70, 3347–3351.
Garel, J.-R., Nall, B. T., and Baldwin, R. L. (1976).Proc. Natl. Acad. Sci. USA 73, 1853–1857.
Grathwohl, C., and Wüthrich, K. (1981).Biopolymers 20, 2623–2633.
Hammes, G. G., and Walz, F. G., Jr. (1969).J. Am. Chem. Soc. 91, 7179–7186.
Houry, W. A., Rothwarf, D. M., and Scheraga, H. A. (1994).Biochemistry (in press).
Howlin, B., Harris, G. W., Moss, D. S., and Palmer, R. A., (1987).J. Mol. Biol. 196, 159–164.
Johnson, M. L., and Faunt, L. M. (1992).Meth. Enzymol. 210, 1–37.
Krebs, H., Schmid, F. X., and Jaenicke, R. (1983).J. Mol. Biol. 169, 619–635.
Kuwajima, K., Mitani, M., and Sugai, S. (1989).J. Mol. Biol. 206, 547–561.
Laity, J. H., Shimotakahara, S., and Scheraga, H. A. (1993).Proc. Natl. Acad. Sci. USA 90, 615–619.
Lewis, P. N., Momany, F. A., and Scheraga, H. A. (1973).Biochem. Biophys. Acta. 303, 211–229.
Lin, L.-N., and Brandts, J. F. (1983).Biochemistry 22, 564–573.
Lin, L.-N., and Brandts, J. F. (1984).Biochemistry 23, 5713–5723.
Lin, L.-N., and Brandts, J. F. (1987).Biochemistry 26, 3537–3543.
Lin, S. H., Konishi, Y., Nall, B. T., and Scheraga, H. A. (1985).Biochemistry 24, 2680–2686.
Mui, P. W., Konishi, Y., and Scheraga, H. A. (1985).Biochemistry 24, 4481–4489.
Oivanen, M., and Lönnberg, H. (1989).J. Org. Chem. 54, 2556–2560.
Pace, C. N., Shirley, B. A., and Thomson, J. A. (1989). InProtein Structure (Creighton, T. E., ed.), IRL Press, Oxford, pp. 311–330.
Press, W. H., Flannery, B. P., Teukolsky, S. A., and Vetterling, W. T. (1990). InNumerical Recipes in C, Cambridge University Press, Oxford, pp. 542–547.
Raleigh, D. P., Evans, P. A., Pitkeathly, M., and Dobson, C. M. (1992).J. Mol. Biol. 228, 338–342.
Rehage, A., and Schmid, F. X. (1982).Biochemistry 21, 1499–1505.
Rothwarf, D. M., and Scheraga, H. A. (1993).Biochemistry 32, 2671–2679.
Sancho, J., Meiering, E. M., and Fersht, A. R. (1991).J. Mol. Biol. 221, 1007–1014.
Schmid, F. X. (1982).Eur. J. Biochem. 128, 77–80.
Schmid, F. X. (1986).Meth. Enzymol. 131, 70–82.
Schmid, F. X., and Blaschek, H. (1981).Eur. J. Biochem. 114, 111–117.
Schultz, D. A., Schmid, F. X., and Baldwin, R. L. (1992).Protein Sci. 1, 917–924.
Segawa, S.-I., and Sugihara, M. (1984).Biopolymers 23, 2489–2498.
Sela, M., and Anfinson, C. B. (1957).Biochim. Biophys. Acta 24, 229–235.
Stimson, E. R., Montelione, G. T., Meinwald, Y. C., Rudolph, R. K. E., and Scheraga, H. A. (1982).Biochemistry 21, 5252–5262.
Sugawara, T., Kuwajima, K., and Sugai, S. (1991).Biochemistry 30, 2698–2706.
Thannhauser, T. W., and Scheraga, H. A. (1985).Biochemistry 24, 7681–7688.
Thannhauser, T. W., Konishi, Y., and Scheraga, H. A. (1987).Meth. Enzymol. 143, 115–119.
Voet, D., Gratzer, W. B., Cox, R. A., and Doty, P. (1963).Biopolymers 1, 193–208.
Weber, P. C., Salemme, F. R., Lin, S. H., Konishi, Y., and Scheraga, H. A. (1985a).J. Mol. Biol. 181, 453.
Weber, P. C., Sheriff, S., Ohlendorf, D. H., Finzel, B. C., and Salemme, F. R. (1985b).Proc. Natl. Acad. Sci. USA 82, 8473–8477.
Wlodawer, A. (1980).Acta Crystallog. B 36, 1826–1831.
Wodak, S. Y., Liu, M. Y., and Wyckoff, H. W. (1977).J. Mol. Biol. 116, 855–875.
Zimmerman, S. S., and Scheraga, H. A. (1976).Macromolecules 9, 408–416.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dodge, R.W., Laity, J.H., Rothwarf, D.M. et al. Folding pathway of guanidine-denatured disulfide-intact wild-type and mutant bovine pancreatic ribonuclease A. J Protein Chem 13, 409–421 (1994). https://doi.org/10.1007/BF01901697
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01901697