Abstract
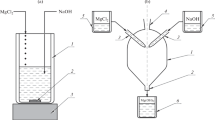
An apparatus is described for studying the velocity of the magnesium reduction process of zirconium production by the “floating-crucible” method, in which the rise in level of the molten MgCl2 + KCl, due to the increase in weight of the reaction crucible immersed in the melt, was measured by means of a Co60 γ-level gauge. The pressure of the zirconium vapor in the reduction apparatus was measured by means of an ordinary pressure gauge, connected by a molten tin seal to the space within the reduction apparatus.
Measurements showed that when the vaporizer was heated to 450 °C, the temperature of the zirconium chloride during magnesium reduction did not exceed 330 °C.
The mean velocity of the magnesium process for the reduction of zirconium chloride increases with increase in the temperature of the reaction crucible from 760 to 850 °C more slowly than with increase in the temperature of the vaporizer from 460 to 490 °C. The deciding factors determining the velocity of the magnesium reduction process of zirconium production are the temperature of the vaporizer and the rate of sublimation and pressure of the zirconium chloride vapor, which are dependent on it.
Similar content being viewed by others
Literature cited
S. M. Shelton and E. D. Billing, The Manufacture of Zirconium Sponge, Zirconium and Zirconium Alloys (A. S. M. Cleveland, Ohio, 1953) p. 82.
F. E. Block and A. D. Abraham, J. Electrochem. Soc. 102, No. 6, 311 (1955).
Rights and permissions
About this article
Cite this article
Reshetnikov, F.G., Oblomeev, E.N. Study of the velocity of the magnesium reduction process of zirconium production. The Soviet Journal of Atomic Energy 4, 459–463 (1958). https://doi.org/10.1007/BF01880569
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01880569