Summary
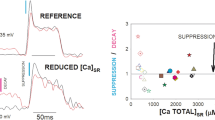
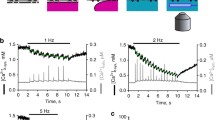
The mechanism of Ca2+ release from the sarcoplasmic reticulum (SR) of slow and fast twitch muscle was compared by examining biochemical characteristics, ryanodine binding. Ca2+ efflux, and single Ca2+ channel properties of SR vesicles. Although many features of the Ca2+ release channel were comparable, two functional assays revealed remarkable differences. The comparable properties include: a high molecular weight protein from both types of muscle was immunologically equivalent, and Scatchard analysis of [3H]ryanodine binding to SR showed that theK d was similar for slow and fast SR. In the flux assay the sensitivity to the agonists caffeine, doxorubicin, and Ca2+ and the antagonists Mg2+, ruthenium red, and tetracaine differed only slightly. When SR vesicles were incorporated into lipid bilayers, the single-channel conductances of the Ca2+ release channels were indistinguishable. The distinguishing properties are: When Ca2+ release from passively45Ca2+-loaded SR were monitored by rapid filtration, the initial rates of Ca2+ release induced by Ca2+ and caffeine were three times lower in slow SR than in fast SR. Similarly, when Ca2+ release channels were incorporated into lipid bilayers, the open probability of the slow SR channel was markedly less, mainly due to a longer mean closed time. Our results indicate that slow and fast muscle have ryanodine receptors that are biochemically analogous, yet functional differences in the Ca2+ release channel may contribute to the different time to peak contraction observed in intact slow and fast muscles.
Similar content being viewed by others
References
Briggs, F.N., Poland, J.L., Solaro, R.J. 1977. Relative capabilities of sarcoplasmic reticulum in fast and slow mammalian skeletal mucles.J. Physiol. 266:587–594
Brody, I.A. 1976. Regulation of isometric contraction in skeletal muscle.Exp. Neurol. 50:673–683
Buller, A.J., Eccles, J.C., Eccles, R.M. 1960. Differentiation of fast and slow muscles in the cat hind limb.J. Physiol. 150:417–439
Campbell, K.P., MacLennan, D.H., Jorgensen, A.O. 1983. Staining of the Ca-binding proteins, calsequestrin, calmodulin, troponin C, and S-100, with the cationic carbocyanine dye “stains-all”.J. Biol. Chem. 258:11267–11273
Close, E.I. 1972. Dynamic properties of mammalian skeletal muscles.Physiol. Rev. 52:129–197
Ehrlich, B.E., Watras, J. 1988. Inositol 1,4,5-trisphosphate activates a channel from smooth muscle sarcoplasmic reticulum.Nature 336:583–586
Endo, M. 1977. Calcium release from the sarcoplasmic reticulum.Physiol. Rev. 57:71–108
Fiehn, W., Peter, J.B. 1971. Properties of the fragmented sarcoplasmic reticulum from fast twich and slow twitch muscles.J. Clin. Invest. 50:570–573
Fitts, R.H., Courtright, J.B., Kim, D.H., Witzmann, F.A. 1982. Muscle fatigue with prolonged exercise: Contractile and biochemical alterations.Am. J. Physiol. 242:C65-C73
Fleischer, S., Ogunbunmi, E.M., Dixon, M.C., Fleer, E.A.M. 1985. Localization of Ca2+ release channels with ryanodine in junctional terminal cisternae of sarcoplasmic reticulum of fast skeletal muscle.Proc. Natl. Acad. Sci. USA.82:7256–7259
Ikemoto, N. 1974. The calcium binding sites involved in the regulation of the purified adenosine triphosphatase of the sarcoplasmic reticulum.J. Biol. Chem. 249:649–651
Ikemoto, N., Ronjat, M., Meszaros, L., Koshita, M. 1989. Postulated role of calsequestrin in the regulation of calcium release from sarcoplasmic reticulum.Biochemistry 28:6764–6771
Imagawa, T., Smith, J. S., Coronado, R., Campbell, K.P. 1987. Purified ryanodine receptor from skeletal muscle sarcoplasmic reticulum is the Ca2+-permeable pore of the calcium release channel.J. Biol. Chem. 262:16636–16643
Imagawa, T., Takasago, T., Shigekawa, M. 1989. Cardiac ryanodine receptor is absent in types I slow skeletal muscle fibers: Immunochemical and ryanodine binding studies.J. Biochem. 106:342–348
Inui, M., Saito, A., Fleischer, S. 1987. Purification of the ryanodine receptor and identity with feet structures of junctional terminal cisternae of sarcoplasmic reticulum from fast skeletal muscle.J. Biol. Chem. 262:1740–1747
Kim, D.H., Ohnishi, S.T., Ikemoto, N. 1983. Kinetics studies of calcium release from sarcoplasmic reticulum in vitro.J. Biol. Chem. 258:9662–9668
Kim, D.H., Witzmann, F.A., Fitts, R.H. 1982. Effect of thyrotoxicosis on sarcoplasmic reticulum in rat skeletal muscle.Am. J. Physiol. 243:C151-C155
Kranias, E.G., Schwartz, A., Jungmann, R. 1982. Characterization of the cyclic 3′, 5′-AMP-dependent protein kinase in sarcoplasmic reticulum and cytosol of canine myocardium.Biochim. Biophys. Acta 709:28–37
Lai, F.A., Erickson, H.P., Rousseau, E., Liu, Q.-Y., Meissner, G. 1988. Purification and reconstitution of the calcium release channel from skeletal muscle.Nature 331:315–319
Lattanzio, F.A., Schlatterer, R.G., Jr., Nicar, M., Campbell, K.P., Sutko, J.L. 1987. The effects of ryanodine on passive calcium fluxes across sarcoplasmic reticulum.J. Biol. Chem. 262:2711–2718
Lowry, O.H., Rosenbrough, N.J., Farr, A.L., Randall, R.J., 1951. Protein measurements with Folin phenol reagent.J. Biol. Chem. 193:265–275
MacLennan, D.H., Brandl, C.J., Korczak, B., Green, N.M. 1985. Amino-acid sequence of a Ca2−+Mg2−-dependent ATPase from rabbit muscle sarcoplasmic reticulum, deduced from its complementary DNA sequence.Nature 316:696–700
Michalak, M., Dupraz, P., Shoshan-Barmatz, V. 1988. Ryanodine binding to sarcoplasmic reticulum membrane: comparison between cardiac and skeletal muscle.Biochim. Biophys. Acta 939:587–594
Mueller, P., Rudin, D.O., Tien, H.T., Wescott, W.C. 1963. Methods for the incorporation of single bimolecular lipid membranes in aqueous solutions.J. Phys. Chem. 67:534–535
Otsu, K., Willard, H.F., Khanna, V.K., Zorzato, F., Green, N.M., MacLennan, D.H. 1990. Molecular cloning of cDNA encoding the Ca2+ release channel (ryanodine receptor) of rabbit cardiac muscle sarcoplasmic reticulum.J. Biol. Chem. 265:13472–13483
Pessah, J.N., Francini, A.O., Scales, D.J., Waterhouse, A.L., Casida, J.E. 1986. Calcium-ryanodine complex: Solubilization and partial characterization from skeletal muscle junctional sarcoplasmic reticulum vesicles.J. Biol. Chem. 261:8643–8648
Peter, J.B., Barnard, J., Edgerton, V.R., Gillespie, C.A. Stempel, K. 1972. Metabolic profiles of three fiber types of skeletal muscle in guinea pigs and rabbits.Biochemistry 11:2627–2633
Rousseau, E., Ladine, J., Liu, O.-Y., Meissner, G. 1988. Activation of the Ca2− release channel of skeletal muscle sarcoplasmic reticulum by caffeine and related compounds.Arch. Biochem. Biophys. 267:75–86
Salviati, G., Volpe, P. 1988. Ca2+ release from sarcoplasmic reticulum of skinned fast- and slow-twitch muscle fibers.Am. J. Physiol. 254:C459-C465
Salviati, G., Volpe, P., Salvatori, S., Betto, R., Damiani, E., Margreth, A., Pasquali-Ronchetti, I. 1982. Biochemical heterogeneity of skeletal muscle microsomal membranes: Membrane origin, membrane specificity and fiber types.Biochem. J. 202:289–301
Smith, J., Coronado, R., Meissner, G. 1986. Single channel measurements of the calcium release channel from skeletal muscle sarcoplasmic reticulum. Activation by calcium and ATP and modulation by magnesium.J. Gen. Physiol. 88:573–588
Sreter, F.A. 1970. Effect of denervaton on fragmented sarcoplasmic reticulum of white and red muscle.Exp. Neurol. 29:52–64
Sreter, F.A., Seidel, J.C., Gergely, J. 1966. Studies on myosin from red and white skeletal muscles of the rabbit. I. Adenosine triphosphatase activity.J. Biol. Chem. 241:5772–5776
Wiehrer, W., Pette, D. 1983. The ratio between intrinsic 115 kDa and 30 kDa peptides as a marker of fibre type-specific sarcoplasmic reticulum in mammalian muscles.FEBS Lett. 158:317–320
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, Y.S., Ondrias, K., Duhl, A.J. et al. Comparison of calcium release from sarcoplasmic reticulum of slow and fast twitch muscles. J. Membrain Biol. 122, 155–163 (1991). https://doi.org/10.1007/BF01872638
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01872638