Summary
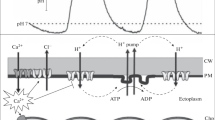
In cells of the freshwater algaHydrodictyon africanum, in solutions where [K+]0=0.1mm and pH0>7.0, the membrane in the light is hyperpolarized. The membrane potential difference {ie179-1} has values from −180 to −275 mV, more negative than any ion diffusion potential difference, and is predominantly a function of pH0, and independent of [K+]0. The hyperpolarization of the membrane appears to arise from an electrogenic efflux of H+, estimated from voltage-clamp data to be about 8 nmol m−2 sec−1 when pH0=8.5. In the light the membrane conductanceg m is about 0.084 S m−2. At light-off, {ie179-2} becomes less negative, with a halftime for change of 15 to 30 sec andg m decreases by about 0.052 S m−2. After dark periods of up to 300 sec, {ie179-3} is largely independent of pH0 for values greater than 6.0 and usually behaves as a combined K+ and Na+ diffusion potential with permeability ratioP Na/P K=0.05 to 0.2. The membrane potassium conductanceg K has either a low value of 2–6×10−2 Sm−2, or a high value of up to 18×10−2 S m−2 depending on [K+]0, the transition from low to high values occurring when {ie179-4} moves over a threshold value that is more negative than {ie179-5}, the electrochemical equilibrium potential for K+. The time for half-change of the transition is about 30 sec. The results are consistent with a model of the membrane in which the pump electromotive force and conductance are in parallel with diffusive electromotive forces and conductances. When the pump is operating its properties determine membrane properties, and when it is inoperative, or running at a diminished rate, the membrane properties are determined more by the diffusive pathways. Changes in both pump rate andg K can account for a variety of characteristic changes in membrane PD and conductance occurring in response to ligh-dark changes, changes in light intensity, pasage of externally applied electric current across the membrane and changes in ionic constituents of the external medium.
Similar content being viewed by others
References
De Michelis, M.I., Raven, J.A., Jayasuriya, H.D. 1979. Measurement of cytoplasmic pH by the DMO technique inHydrodictyon africanum.J. Exp. Bot. 30:681–695
Felle, H., Bentrup, F.W. 1976. Effect of light upon membrane potential, conductance, and ion fluxes inRiccia fluitans.J. Membrane Biol. 27:153–170
Findlay, G.P., Hope, A.B. 1976. Electrical properties of plant cells: Methods and findings.In:Encyclopedia of Plant Physiology, New Series. U. Lüttage and M.G., Pitman, editors. Vol. 2, Part A, pp. 53–92. Springer, Berlin
Hodgkin, A.L., Katz, B. 1949. The effect of sodium ions on the electrical activity of the giant axon of the squid.J. Physiol. (London) 108:33–77
Hope, A.B. 1965. Ionic relations of cells ofChara australis. X. Effects of bicarbonate ions on electrical properties.Aust. J. Biol. Sci. 18:789–801
Hope, A.B., Walker, N.A., 1961. Ionic relations of cells ofChara australis R. Br. IV. Membrane potential differences and resistances.Aust. J. Biol. Sci. 14:26–44
Lucas, W.J. 1977. Analogue inhibition of the active HCO −3 transport site in the characean plasma membrane.J. Exp. Bot. 28:1321–1336
Metlička, R., Rybová, R. 1967. Oscillations of the trans-membrane potential difference in the algaHydrodictyon reticulatum.Biochim. Biophys. Acta 135:563–565
Raven, J.A. 1967. Ion transport inHydrodictyon africanum.J. Gen Physiol. 50:1627–1640
Raven, J.A. 1968. The mechanism of photosynthetic use of bicarbonate byHydrodictyon africanum.J. Exp. Bot. 19:193–206
Rybová, R., Janacek, K., Slavikova, M. 1977. Electrical and pH transients in the algaHydrodictyon reticulatum.In: Transmembrane Ionic Exchanges in Plants. M. Thellier, A. Monnier and J. Dainty, editors. pp. 357–364.CNRS, Paris
Smith, F.A., Raven, J.A. 1979. Intracellular pH and its regulation.Annu. Rev. Plant Physiol. 30:289–311
Smith, F.A., Raven, J.A., Jayasuriya, H.D. 1978. Uptake of methyl-ammonium ions byHydrodictyon africanum.J. Exp. Bot. 29:121–133
Spanswick, R.M. 1970. The effects of bicarbonate ions and external pH on the membrane potential and resistance ofNitella translucens.J. Membrane Biol. 2:59–70
Spanswick, R.M. 1973. Electrogenesis in photosynthetic tissues.In: Ion Transport in Plants. W.P. Anderson, editor. pp. 113–128. Academic Press, London
Spanswick, R.M. 1981. Electrogenic ion pumps.Annu. Rev. Plant Physiol. 32:267–289
Walker, N.A., Smith, F.A. 1975. Intracellular pH inChara corallina measured by DMO distribution.Plant Sci. Lett. 4: 125–132
Walker, N.A., Smith, F.A. 1977. Circulating electric currents between acid and alkaline zones associated with HCO −3 assimilation inChara.J. Exp. Bot. 28:1190–1206
Walker, N.A., Smith, F.A., Cathers, I.R., 1980. Bicarbonate assimulation by fresh-water charophytes and higher plants. I. Membrane transport of bicarbonate ions is not proven.J. Membrane Biol. 57:51–58
Wyn Jones, R.G., Brady, C.J., Speirs, J., 1979. Ionic and osmotic relations in plant cells.In: Recent Advances in the Biochemistry of Cereals. D.L. Laidman and R.G. Wyn Jones, editors. pp. 61–103. Academic Press, London, New York, San Francisco
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Findlay, G.P. Electrogenic and diffusive components of the membrane ofHydrodictyon africanum . J. Membrain Biol. 68, 179–189 (1982). https://doi.org/10.1007/BF01872263
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01872263