Summary
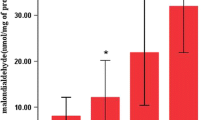
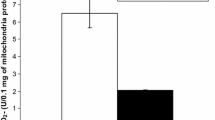
Reduced glutathione (GSH) protects alveolar macrophages (AMs) and polymorphonuclear leukocytes (PMNs) against oxidative damage. To obtain further knowledge of the oxygen toxicity we determined GSH in AMs and PMNs of guinea pigs exposed to an oxygen concentration of 85% for up to 90 h. AMs and PMNs from control animals contained 17.93 and 11.67 nmol GSH/mg protein, respectively. During the exposure to a FIO2 of 85% we observed a significant continuous increase of GSH in AMs. By 90 h of oxygen exposure, AMs contained 51.22 nmol GSH/mg protein. In addition, the protein content of AMs decreased during hyperoxia. In contrast, no change of the GSH amount and protein content was detectable in PMNs. The increase of GSH in AMs could serve as an adaptation of the cells to hyperoxia. The lack of the GSH increase in PMNs could be due to the different oxygen concentrations between the lung and the peritoneal cavity. The greater GSH content in AMs may account for the difference between these cells in their susceptibility to oxidant injury.
Similar content being viewed by others
References
Beutler E (1971) Abnormalities of the hexose monophosphate shunt. Semin Hematol 8:311–320
Beutler E, Duron O, Kelly BM (1963) Improved method for the determination of blood glutathione. J Lab Clin Med 61:882–888
Bowden DH, Adamson IYR (1974) Endothelial regeneration as a marker of the differential vascular responses in oxygen induced pulmonary edema. Lab Invest 30:350–357
Clark JM (1974) The toxicity of oxygen. Rev Respir Dis 110:40–50
Crapo JD, Tierney DF (1974) Superoxide dismutase and pulmonary oxygen toxicity. Am J Physiol 226:1401–1407
Flohé L (1971) Die Glutathionperoxidase: Enzymologie und biologische Aspekte. Klin Wochenschr 12:669–683
Flohé L, Günzler WA (1974) Glutathione peroxidase. In: Flohé L, Benöhr HC, Sies H, Waller HD, Wendel A (eds) Glutathione. Thieme, Stuttgart, pp 132–143
Flohé L, Günzler WA (1976) Glutathione-dependent enzymatic oxidoreduction reaction. In: Arias IM, Jakoby WB (eds) Glutathione, metabolism and function. Raven Press, New York, pp 17–34
Fridovich I (1970) Quantitative aspects of the production of superoxide anion radical by milk xanthine oxidase. J Biol Chem 245:4053–4057
Fridovich I (1975) Oxygen: Boon and bane. Am Sci 63:54–59
Gregory EM, Fridovich I (1973) Oxygen toxicity and the superoxide dismutase. J Bacteriol 114:1193–1197
Hartter P, Weber U (1974) The thiol-disulfide exchange reactions of asymmetric disulfides of cysteine and cyclic cysteine peptides with GSH. In: Flohé L, Benöhr HC, Sies H, Waller HD, Wendel A (eds) Glutathione. Thieme, Stuttgart, pp 29–33
Huber GL, Finder E, La Force FM (1972) Prevention of oxygen toxicity in the lung. Chest 62:365–371
Immich H (1974) Medizinische Statistik. Schattauer, Stuttgart New York
Kellogg EW, Fridovich I (1975) Superoxide, hydrogen peroxide, and singlet oxygen in lipid peroxidation by a xanthine oxidase system. J Biol Chem 250:8812–8817
Kimball RE, Reddy K, Pierce TH, Schwartz LW, Mustafa MG, Gross CE (1976) Oxygen toxicity: Augmentation of antioxidant defense mechanisms in rat lung. Am J Physiol 230:1425–1431
Kosower EM (1976) Chemical properties of glutathione. In: Arias IM, Jakoby WB (eds) Glutathione, metabolism and function. Raven Press, New York, pp 1–15
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–274
Oliver JM, Albertini DF, Berlin RD (1976) Effects of glutathione-oxidizing agents on microtubule assembly and microtubule-dependent surface properties of human neutrophils. J Cell Biol 71:921–932
Oren R, Farnham AE, Saito K, Milowsky E, Karnovsky ML (1963) Metabolic patterns in three types of phagocytizing cells. J Cell Biol 17:487–501
Pratt PC (1974) Pathology of pulmonary oxygen toxicity. Am Rev Respir Dis 110:51–57
Resnick JS, Brown DM, Vernier RL (1974) Oxygen toxicity in fetal organ culture. II. The developing lung. Lab Invest 31:665–677
Rister M, Baehner RL (1975) Induction of superoxide dismutase in polymorphonuclear leukocytes and alveolar macrophages. Blood 46:1015
Rister M, Baehner RL (1976) The alteration of superoxide dismutase, catalase, glutathione peroxidase and NAD(P)H cytochrome C reductase in guinea-pig polymorphonuclear leukocytes and alveolar macrophages during hyperoxia. J Clin Invest 58:1174–1184
Rister M, Baehner RL (1977) Effects of hyperoxia on superoxide anion and hydrogen peroxide production of polymorphonuclear leukocytes and alveolar macrophages. Br J Haematol 36:241–248
Rister M, Baehner RL (1978) Neue Aspekte zur Toxizität des Sauerstoffs. Dtsch Med Wochenschr 103:977–981
Rister M (1982) Effects of hyperoxia on phagocytosis. Blut 45:157–166
Roos D, Weening RS, Voetman AA, van Schaik MLJ, Bot AAM, Meerhof LJ, Loos JA (1979) Protection of phagocytic leukocytes by endogenous glutathione: Studies in a family with glutathione reductase deficiency. Blood 53:851–866
Saltzman HA, Fridovich I (1973) Oxygen toxicity. Introduction to a protective enzyme: Superoxide dismutase. Circulation 48:921–923
Stankova L, Rigas DA, Keown P, Bigley R (1977) Leukocyte ascorbate and glutathione: Potential capacity for inactivating oxidants and free radicals. J Reticuloendothel Soc 21:97–102
Yam J, Frank L, Roberts RJ (1978) Oxygen toxicity: Comparison of lung biochemical responses in neonatal and adult rats. Pediatr Res 12:115–119
Author information
Authors and Affiliations
Additional information
Dedicated to Prof. Dr. E. Gladtke on the occasion of his 60th birthday
Supported by the Deutsche Forschungsgemeinschaft (project no. 275/5)
This study contains data of a thesis submitted in partial fulfilment of the requirement for the degree of a Medical Doctor
Rights and permissions
About this article
Cite this article
Rister, M., Wustrow, C. Effect of hyperoxia on reduced glutathione in alveolar macrophages and polymorphonuclear leukocytes. Res. Exp. Med. 185, 445–450 (1985). https://doi.org/10.1007/BF01851850
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01851850