Summary
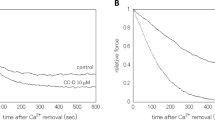
The role of protein kinase C (PKC) in regulating the contractile state of smooth muscle was investigated using the constitutively active catalytic fragment of PKC (PKM) with skinned (demembranated) chicken gizzard fibres. PKM attenuated a submaximal contraction in gizzard smooth muscle skinned fibres, but not in rabbit cardiac skinned fibres. PKM-mediated relaxation of submaximal contractions of smooth muscle was accompanied by a reduction in the rate of ATP hydrolysis in the fibre and by phosphorylation of the 20 kDa light chain of gizzard myosin at the PKC sites (serine-1, serine-2 and threonine-9). In addition, several other endogenous proteins were phosphorylated by PKM. However, the inhibitory effects on tension and ATPase are consistent with the biochemical effects of PKC-catalysed phosphorylation of Myosin, I.e. reduction of the actin-activated MgATPase activity of myosin prephosphorylated at serine-19 by myosin light chain kinase. Pretreatment of skinned fibres with PKM and ATPγS in the absence of Ca2+ had no inhibitory effect on the subsequent submaximal Ca2+-activation of force. Consistent with this observation, PKC was not able to utilize ATPγS as a substrate, confirming that the observed effects were the result of PKM-catalysed protein phosphorylation. We suggest that PKC may have two distinct effects on smooth muscle contraction: translocation of PKC to the sarcolemma on stimulation results in phosphorylation of a protein(s) other than myosin and a slow, sustained contraction; in some circumstances PKC may undergo proteolysis to PKM resulting in myosin phosphorylation at PKC-specific sites, a reduction in ATPase activity and relaxation of the muscle.
Similar content being viewed by others
Abbreviations
- Ap5A:
-
P1, P5-di(adenosine-5′)pentaphosphate
- ATPγS:
-
adenosine-5′-O-(3-thiotriphosphate)
- TPA:
-
12-O-tetradecanoylphorbol-13-acetate
- TPCK:
-
L-1-tosylamido-2-phenylethyl chloromethyl ketone
References
Baraban, J. M., Gould, R. J., Peroutka, S. J. &Snyder, S. H. (1985) Phorbol ester effects on neurotransmission: interaction with neurotransmitters and calcium in smooth muscle.Proc. Natl. Acad. Sci. USA 82, 604–7.
Bell, R. M., Hannun, Y. &Loomis, C. (1986) Mixed micelle assay of protein kinase C.Methods Enzymol. 124, 353–9.
Bengur, A. R., Robinson, E. A., Appella, E. &Sellers, J. R. (1987) Sequence of the sites phosphorylated by protein kinase C in the smooth muscle myosin light chain.J. Biol. Chem. 262, 7613–7.
Cassidy, P., Hoar, P. E. &Kerrick, W. G. L. (1979) Irreversible thiophosphorylation and activation of tension in functionally skinned rabbit ileum strips by [35S]ATPγS.J. Biol. Chem. 254, 11148–53.
Chatterjee, M. &Tejada, M. (1986) Phorbol ester-induced contraction in chemically skinned vascular smooth muscle.Am. J. Physiol. 251, C356–61.
Chida, K., Kato, N. &Kuroki, T. (1986) Down regulation of phorbol diester receptors by proteolytic degradation of protein kinase C in a cultured cell line of fetal rat skin keratinocytes.J. Biol. Chem. 261, 13013–18.
Colburn, J. C., Michnoff, C. H., Hsu, L. C., Slaughter, C. A., Kamm, K. E. &Stull, J. T. (1988) Sites phosphorylated in myosin light chain in contracting smooth muscle.J. Biol. Chem. 263, 19166–73.
Danthuluri, N. R. &Deth, R. C. (1984) Phorbol ester-induced contraction of arterial smooth muscle and inhibition ofα-adrenergic response.Biochem. Biophys. Res. Commun. 125, 1103–9.
Endo, T., Naka, M. &Hidaka, H. (1982) Ca2+-phospholipid dependent phosphorylation of smooth muscle myosin.Biochem. Biophys. Res. Commun. 105, 942–8.
Erdodi, F., Barany, M. &Barany, K. (1987) Myosin light chain isoforms and their phosphorylation in arterial smooth muscle.Circ. Res. 61, 898–903.
Erdodi, F., Rokolya, A., Barany, M. &Barany, K. (1988) Phosphorylation of the 20 000-Da myosin light chain isoforms of arterial smooth muscle by myosin light chain kinase and protein kinase C.Arch. Biochem. Biophys. 266, 583–91.
Fabbro, D., Regazzi, R., Costa, S. D., Borner, C. &Eppenberger, U. (1986) Protein kinase C desensitization by phorbol esters and its impact on growth of human breast cancer cells.Biochem. Biophys. Res. Commun. 135, 65–73.
Griffiths, P. J., Güth, K., Kuhn, H. J. &Rüegg, J. C. (1980) ATPase activity in rapidly activated skinned muscle fibres.Pflügers Archiv. 387, 167–73.
Güth, K. &Wojciechowski, R. (1986) Perfusion cuvette for the simultaneous measurement of mechanical, optical and energetic parameters of skinned muscle fibres.Pflügers Archiv. 407, 552–7.
Guy, G. R., Gordon, J., Walker, L., Michell, R. H. &Brown, G. (1986) Redistribution of protein kinase C during mitogenesis of human B lymphocytes.Biochem. Biophys. Res. Commun. 135, 146–53.
Haller, H., Smallwood, J. I. &Rasmussen, H. (1990) Protein kinase C translocation in intact vascular smooth muscle strips.Biochem. J. 270, 375–81.
Hartshorne, D. J. (1987) Biochemistry of the contractile process in smooth muscle. InPhysiology of the Gastrointestinal Tract. Second Edition (edited by Johnson, L. R.) pp. 423–82. New York: Raven Press.
Hathaway, D. R. &Haeberle, J. R. (1983) Selective purification of the 20 000-Da light chains of smooth muscle myosin.Anal. Biochem. 135, 37–43.
Hellam, D. C. &Podolsky, R. J. (1969) Force measurements in skinned muscle fibres.J. Physiol. 200, 807–19.
Hoar, P. E., Kerrick, W. G. L. &Cassidy, P. S. (1979) Chicken gizzard: relation between calcium-activated phosphorylation and contraction.Science 204, 503–6.
Hoar, P. E., Pato, M. D. &Kerrick, W. G. L. (1985) Myosin light chain phosphatase. Effect on the activation and relaxation of gizzard smooth muscle skinned fibres.J. Biol. Chem. 260, 8760–4.
Hoar, P. E., Potter, J. D. &Kerrick, W. G. L. (1988) Skinned ventricular fibres: troponin C extraction is species-dependent and its replacement with skeletal troponin C changes Sr2+activation properties.J. Muscle. Res. Cell Motil. 9, 165–73.
Huang, K.-P. &Huang, F. L. (1986) Conversion of protein kinase C from a Ca2+-dependent to an independent form of phorbol ester-binding protein by digestion with trypsin.Biochem. Biophys. Res. Commun. 139, 320–6.
Ikebe, M., Hartshorne, D. J. &Elzinga, M. (1987) Phosphorylation of the 20 000-Dalton light chain of smooth muscle myosin by the calcium-activated, phospholipid-dependent protein kinase. Phosphorylation sites and effects of phosphorylation.J. Biol. Chetn. 262, 9569–73.
Inagaki, M., Yokokura, H., Itoh, T., Kanmura, Y., Kuriyama, H. &Hidaka, H. (1987) Purified rabbit brain protein kinase C relaxes skinned vascular smooth muscle and phosphorylates myosin light chain.Arch. Biochem. Biophys. 254, 136–41.
Itoh, T., Kanmura, Y., Kuriyama, H. &Sumimoto, K. (1986) A phorbol ester has dual actions on the mechanical response in the rabbit mesenteric and porcine coronary arteries.J. Physiol. 375, 515–34.
Jiang, M. J. &Morgan, K. G. (1987) Intracellular calcium levels in phorbol ester-induced contractions of vascular muscle.Am. J. Physiol. 253, H1365–71.
Jiang, M. J. &Morgan, K. G. (1989) Agonist-specific myosin phosphorylation and intracellular calcium during isometric contractions of arterial smooth muscle.Pflügers Arch. 413, 637–43.
Kamm, K. E., Hsu, L. C., Kubota, Y. &Stull, J. T. (1989) Phosphorylation of smooth muscle myosin heavy and light chains. Effects of phorbol dibutyrate and agonists.J. Biol Chem. 264, 21223–9.
Kenney, R. E., Hoar, P. E. &Kerrick, W. G. L. (1990) The relationship between ATPase activity, isometric force, and myosin light-chain phosphorylation and thiophosphorylation in skinned smooth muscle fibre bundles from chicken gizzard.J. Biol. Chem. 265, 8642–9.
Kishimoto, A., Kajikawa, N., Shiota, M. &Nishizuka, Y. (1983) Proteolytic activation of calcium-activated, phospholipid-dependent protein kinase by calcium-dependent neutral protease.J. Biol. Chem. 258, 1156–64.
Klee, C. B. (1977) Conformational transition accompanying the binding of Ca2+ to the protein activator of 3′,5′-cyclic adenosine monophosphate phosphodiesterase.Biochemistry 16, 1017–24.
Laemmli, U. K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4.Nature 227, 680–5.
Melloni, E., Pontremoli, S., Michetti, M., Sacco, O., Spara-Tore, B. &Horecker, B. L. (1986) Involvement of calpain in the activation of protein kinase C in neutrophils stimulated by phorbol myristic acid.J. Biol. Chem. 261, 4101–5.
Menkes, H., Baraban, J. M. &Snyder, S. H. (1986) Protein kinase C regulates smooth muscle tension in guinea pig trachea and ileum.Eur. J. Pharmacol. 122, 19–27.
Murachi, T., Tanaka, K., Hatanaka, M. &Muramaki, T. (1981) Intracellular Ca2+-dependent protease (calpain) and its high-molecular-weight endogenous inhibitor (calpastatin).Adv. Enzyme Regul. 19, 407–24.
Ngai, P. K., Carruthers, C. A. &Walsh, M. P. (1984) Isolation of the native form of chicken gizzard myosin light chain kinase.Biochem. J. 218, 863–70.
Nishikawa, M., Hidaka, H. &Adelstein, R. S. (1983) Phosphorylation of smooth muscle heavy meromyosin by calciumactivated, phospholipid-dependent protein kinase. The effect on actin-activated MgATPase activity.J. Biol. Chem. 258, 14069–72.
Nishikawa, M., Sellers, J. R., Adelstein, R. S. &Hidaka, H. (1984) Protein kinase C modulatesin vitro phosphorylation of the smooth muscle heavy meromyosin by myosin light chain kinase.J. Biol. Chem. 259, 8808–14.
Nishizuka, Y. (1986) Studies and perspectives of protein kinase C.Science 233, 305–12.
Okamoto, Y. &Sekine, T. (1978) Effects of tryptic digestion on the enzymatic activities of chicken gizzard myosin.J. Biochem. (Tokyo) 83, 1375–9.
Park, S. &Rasmussen, H. (1985) Activation of tracheal smooth muscle contraction: synergism between Ca2+ and activators of protein kinase C.Proc. Natl. Acad. Sci. USA 82, 8835–9.
Pearson, R. B., Jakes, R., John, M., Kendrick-Jones, J. &Kemp, B. E. (1984) Phosphorylation site sequence of smooth muscle myosin light chain (Mr=20 000).FEBS Lett. 168, 108–12.
Persechini, A. &Hartshorne, D. J. (1981) Phosphorylation of smooth muscle myosin: evidence for cooperativity between the myosin heads.Science 213, 1383–5.
Pontremoli, S., Melloni, E., Sparatore, B., Michetti, M., Salamino, F. &Horecker, B. L. (1990a) Isozymes of protein kinase C in human neutrophils and their modification by two endogenous proteinases.J. Biol. Chem. 265, 706–12.
Pontremoli, S., Michetti, M., Melloni, E., Sparatore, B., Salamino, F. &Horecker, B. L. (1990b) Identification of the proteolytically activated form of protein kinase C in stimulated human neutrophils.Proc. Natl. Acad. Sci. USA 87, 3705–7.
Rasmussen, H., Forder, J., Kojima, I. &Scriabine, A. (1984) TPA-induced contraction of isolated rabbit vascular smooth muscle.Biochem. Biophys. Res. Commun. 122, 776–84.
Sellers, J. R., Pato, M. D. &Adelstein, R. S. (1981) Roles of calcium and phosphorylation in the regulation of the activity of gizzard myosin.J. Biol. Chem. 256, 13137–42.
Sherry, J. M. F., Gorecka, A., Aksoy, M. O., Dabrowska, R. &Hartshorne, D. J. (1978) Reversible phosphorylation of smooth muscle myosin, heavy meromyosin, and platelet myosin.Biochemistry 17, 4411–18.
Singer, H. (1990) Protein kinase C activation and myosin light chain phosphorylation in32P-labelled arterial smooth muscle.Am. J. Physiol. 259, C631–9.
Singer, H. A. &Baker, K. M. (1987) Calcium dependence of phorbol 12,13-dibutyrate-induced force and myosin light chain phosphorylation in arterial smooth muscle.J. Pharmacol. Exp. Ther. 243, 814–21.
Singer, H. A., Oren, J. W. &Benscoter, H. A. (1989) Myosin light chain phosphorylation in32P-labelled rabbit aorta stimulated by phorbol 12,13-dibutyrate and phenylephrine.J. Biol. Chem. 264, 21215–22.
Spector, T. (1978) Refinement of the Coomassie Blue method of protein quantitation. A simple and linear spectrophotometric assay for ≤ 0.5 to 50 μ of protein.Anal. Biochem. 86, 142–6.
Sutton, T. A. &Haeberle, J. R. (1990) Phosphorylation by protein kinase C of the 20 000-Dalton light chain of myosin in intact and chemically skinned vascular smooth muscle.J. Biol. Chem. 265, 2749–54.
Takashi, R. &Putnam, S. (1979) A fluorimetric method for continuously assaying ATPase: application to small specimens of glycerol-extracted muscle fibres.Anal. Biochem. 92, 375–82.
Tapley, P. M. &Murray, A. W. (1984) Modulation of Ca2+-activated, phospholipid-dependent protein kinase in platelets treated with a tumour-promoting phorbol ester.Biochem. Biophys. Res. Commun. 112, 158–64.
Tapley, P. M. &Murray, A. W. (1985) Evidence that treatment of platelets with phorbol ester causes proteolytic activation of Ca2+-activated, phospholipid-dependent protein kinase.Eur. J. Biochem. 151, 419–23.
Walsh, M. P., Bridenbaugh, R., Hartshorne, D. J. &Kerrick, W. G. L. (1982) Phosphorylation-dependent activated tension in skinned gizzard muscle fibres in the absence of Ca2+.J. Biol. Chem. 257, 5987–90.
Walsh, M. P., Hinkins, S., Dabrowska, R. &Hartshorne, D. J. (1983) Purification of smooth muscle myosin light chain kinase.Methods Enzymol. 99, 279–88.
Walsh, M. P., Valentine, K. A., Ngai, P. K., Carruthers, C. A. &Hollenberg, M. D. (1984) Ca2+-dependent hydrophobicinteraction chromatography. Isolation of a novel Ca2+-binding protein and protein kinase C from bovine brain.Biochem. J. 244, 117–27.
Wolf, M., Cuatrecasas, P. &Sahyoun, N. (1985) Interaction of protein kinase C with membranes is regulated by Ca2+, phorbol esters and ATP.J. Biol. Chem. 260, 15718–22.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Parente, J.E., Walsh, M.P., Kerrick, W.G.L. et al. Effects of the constitutively active proteolytic fragment of protein kinase C on the contractile properties of demembranated smooth muscle fibres. J Muscle Res Cell Motil 13, 90–99 (1992). https://doi.org/10.1007/BF01738432
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01738432