Summary
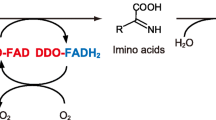
Partially purifiedD-aspartate oxidase from beef kidney has been tested in the presence of butanedione or phenylglyoxal, which specifically modify the arginine molecule. The results obtained clearly indicate that arginine residues are involved in the binding of the substrate to the active site of the enzyme.
Similar content being viewed by others
References
Riordan, J. F. and Scandurra, R., 1975. Biochem. Biophys. Res. Comm. 66, 417–424.
Riordan, J. F., 1973. Biochemistry 12, 3915–3923.
Takahashi, K., 1968. J. Biol. Chem. 243, 6171–6179.
Rinaldi, A., 1971. Enzymologia 40, 314–321.
Duley, J. and Holmes, R. S., 1975. Anal. Biochem. 69, 164–169.
Lange, L. G., Riordan, J. F. and Vallee, B. L., 1974. Biochemistry 13, 4361–4370.
David, M., Rasched, I. and Sund, H., 1976. FEBS Letters 62, 288–292.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Crifò, C., Santoro, L., Rinaldi, A. et al. Essential arginine residues in beef kidney D-aspartate oxidase (a preliminary report). Mol Cell Biochem 17, 7–9 (1977). https://doi.org/10.1007/BF01732548
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01732548