Summary
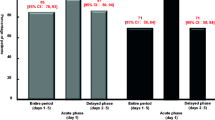
Twenty nonseminomatous testicular cancer patients not pretreated with emetogenic chemotherapy were included in a crossover study of antiemetic therapy. Patients were randomly assigned to receive either nabilone (2×2 mg/day) or alizapride (3×150 mg/day) prior to beginning lowdose cisplatin chemotherapy. Patients on nabilone had significantly fewer episodes of emesis than those on alizapride (medians, 1.1 vs 2.9;p<0.01). Nabilone was superior to alizapride in giving complete relief from nausea (medians, 65% vs 30%;p<0.01), and was more effective in shortening the duration of nausea (medians, 1.3 h vs 5.1 h;p<0.01); however, it caused more adverse effects. It is concluded that nabilone has greater antiemetic activity than alizapride in young patients receiving low-dose cisplatin chemotherapy. Nabilone dosage should be reduced to decrease the incidence and degree of adverse reactions while leaving the definite antiemetic activity unchanged.
Similar content being viewed by others
References
Archer AR, Blanchard WB, Day WA, Johnson DW, Lavaganino ER, Ryan CW (1977) Cannabinoids. 3. Synthetic approaches to 9-ketocannabinoids. Total synthesis of nabilone. J Org Chem 42:2277–2284
Bleiberg H, Rozencweig M, Nicaise C (1983) Phasc I study of alizapride, a new antiemetic agent. 2nd European Conference on Clinical Oncology and Cancer Nursing, Amsterdam, Abstracts, 219 (19–43)
Cheymol G, Mouillé P (1982) Etude des effects cardiovasculaires et anti-arythmisants de l'alizapride. Sem Hop Paris 58:333–338
Cupissol D, Favier F, Favier C, Serrou B (1982) Evaluation d'un nouvel anti-emetic l'alizapride, en cancérologie. Sem Hop Paris 58:1370–1373
Gralla RJ, Tyson LB, Bordin LA, Clark RA, Kelsen DP, Kris MG, Kalman LB, Groshen S (1984) Antiemetic therapy: A review of recent studies and a report of a random assignment trial comparing metoclopramide with delta-9-tetrahydrocannabinol. Cancer Treat Rep 68:163–172
Herman TS, Einhorn LH, Jones SE, Nagy C, Chester AB, Dean JC, Furnas B, Williams SD, Leigh SA, Dorr RT, Moon TE (1979) Superiority of nabilone over prochlorperazine as an antiemetic in patients receiving cancer chemotherapy. N Engl J Med 300:1295–1297
Higi M, Niederle N, Bremer K, Schmitt G, Schmidt CG, Seeber S (1982) Levonantradol bei der Behandlung von zytostatikabedingter Übelkeit und Erbrechen. Klinische Erfahrungen. Dtsch med Wochenschr 107:1232–1234
Hutcheon AW, Palmer JBD, Soukop M, Cunningham D, McArdle C, Welsh J, Stuart F, Sangster G, Kaye S, Charlton D, Cash H (1985) A randomised multicentre single blind comparison of a cannabinoid anti-emetic (Levonantradol) with chlorpromazine in patients receiving their first cytotoxic chemotherapy. Eur J Cancer Clin Oncol 19:1087–1090
Johansson R, Kilkku P, Groenroos M (1982) A double-blind, controlled trial of nabilone vs. prochlorperazine for refractory emesis induced by cancer chemotherapy. Cancer Treat Rev [Suppl B] 9:25–33
Jones SE, Durant JR, Greco FA, Robertone A (1982) A multi-institutional phase III study of nabilone vs. placebo in chemotherapy-induced nausea and vomiting. Cancer Treat Rev [Suppl B] 9:45–48
Laville CL, Margarit J (1982) Etude pharmacodynamique de l'alizapride. Sem Hop Paris 58:323–331
Lemberger L, Rowe H (1975) Clinical pharmacology of nabilone, a cannabinol derivative. Clin Pharmacol Ther 18:720–726
Levitt M (1982) Nabilone vs. placebo in the treatment of chemotherapy-induced nausea and vomiting in cancer patients. Cancer Treat Rev [Suppl B] 9:49–53
Manegold C, Abel U, Fritze D, Herrmann R (1983) The antiemetic effect of alizapride upon cisplatin-induced emesis. 2nd European Conference on Clinical Oncology and Cancer Nursing, Amsterdam, Abstracts, 216 (19–27)
Morrow GR (1984) Clinical characteristics associated with the development of anticipatory nausea and vomiting in cancer patients undergoing chemotherapy treatment. J Clin Oncol 2:1170–1176
Perrot J, Nahas G, Laville C, Debay A (1981) Substituted benzamides as antiemetics. In: Poster DS, Penta JS, Bruno S (eds) Treatment of cancer chemotherapy-induced nausea and vomiting. Masson Publishing, New York Paris Barcelona Milan Mexico City Rio de Janeior, pp 195–207
Preusser P, Achterrath W, Niederle N, Seeber S (1985) Cisplatin. Arzneimitteltherapie 3:50–65
Schütte J, Niederle N, Krischke W, Giesche U, Seeber S (1983) Antiemetische Wirksamkeit von Alizaprid bei Cisplatin-bedingtem Erbrechen. Tumordiagnostik & Therapie 4:146–149
Steele N, Gralla RJ, Braun DW, Young CW (1980) Doubleblind comparison of the antiemetic effects of nabilone and prochlorperazine on chemotherapy-induced emesis. Cancer Treat Rep 64:219–224
Wada JK, Bogdon DL, Gunnell JC, Hum GJ, Gota CH, Rieth TE (1982) Double-blind, randomized, crossover trial of nabilone vs. placebo in cancer chemotherapy. Cancer Treat Rev [Suppl B] 9:39–44
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Niederle, N., Schütte, J. & Schmidt, C.G. Crossover comparison of the antiemetic efficacy of nabilone and alizapride in patients with nonseminomatous testicular cancer receiving cisplatin therapy. Klin Wochenschr 64, 362–365 (1986). https://doi.org/10.1007/BF01728184
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01728184