Summary
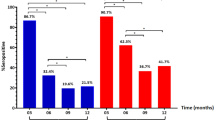
Infants with respiratory syncytial virus (RSV) infection were shown to have antibodies against HEp-2 cell antigen present in RSV-antigen preparation used for immunoblot analysis. The prevalence of anti HEp-2 cell antibodies was examined in infants hospitalized for RSV infection (n=49, median age 121 days) compared to rotavirus infected children (n=30, median age 114 days) and to healthy controls (n=20, median age 150 days). The immunoblot analysis with RSV-infected and non-infected HEp-2 cells as antigen revealed the expected age-dependent low prevalence of G protein antibodies and clear seroconversion of N and P protein antibodies. HEp-2 antibody prevalence was higher in RSV antigen-positive infants (33/49) than in rotavirus antigen-positive (5/30) and RSV antigen-negative infants (4/20), respectively (p<0.001). Anti HEp-2 antibodies were mostly directed against 47, 46, 33, 30 kD antigens. A multiple regression analysis found the following correlations (odds ratio; 95% confidence interval): 42 kD RSV antibodies (N protein) with pneumonia (7.58; 1.43-40), 94 kD RSV antibodies (G protein) with bronchiolitis (0.064; 0.006–0.686). This study shows repeated well-known features of humoral immunity in RSV infection. The data on anti HEp-2 antibodies point to a role for these pre-existing autoreactive antibodies in the pathogenesis of RSV infection.
Zusammenfassung
Bei der Antikörperbestimmung gegen RS-Virus im Immunoblot bei Säuglingen und Kindern, die wegen ihrer Erkrankung hospitalisiert waren, wurden Antikörper gegen HEp 2-Zell-Antigene bei 33 von 49 Kindern (medianes Alter 121 Tage; 67%) gefunden. Die Prävalenz bei gesunden Kontrollen (n=20, medianes Alter 150 Tage, Anti-HEp 2-Zell-Antikörper bei 20%) und bei Kindern, die wegen Rotavirusinfektion hospitalisiert waren (n=30, medianes Alter 114 Tage, Anti-HEp 2-Zell-Antikörper bei 17%), waren signifikant niedriger (p<0,001). Die Anti-HEp 2-Zell-Antikörper reagierten hauptsächlich mit Antigenen von 47, 46, 33, 30 kD. Für die RSV-spezifischen Antikörper wurde in einer multiplen Regressionsanalyse eine Korrelation für N-Protein-Antikörper mit dem Vorhandensein einer Pneumonie (odds ratio; 95% confidence interval: 7,58; 1,43–40) und für das Vorhandensein von G-Protein-Antikörpern mit Bronchiolitis (0,064; 0,006–0,68) gefunden. Insofern zeigt die Untersuchung bekannte Zusammenhänge der spezifischen RSV-Antikörper mit Immunität und Morbidität. Die erhobenen Daten über Anti-HEp 2-Antikörper könnten auf eine Rolle dieser bei Erkrankungsbeginn vorhandenen, autoreaktiven Antikörper in der Pathogenese der RSV-Infektion hinweisen.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aherne, W., Bird, T., Court, S. D., Gardner, P. S., McQuillen, J. Pathological changes in virus infections of the lower respiratory tract in children. J. Clin. Pathol. 23 (1970) 7–18.
Isaacs, D., Bangham, C. R. M., McMichael, A. J. Cell-mediated cytotoxic response to respiratory syncytial virus in infants with bronchiolitis. Lancet ii (1987) 769–771.
Welliver, R. C., Kaul, T. N., Ogra, P. L. The appearance of cell-bound IgE in respiratory-tract epithelium after respiratory-syncytial-virus infection. N. Engl. J. Med. 303 (1980) 1198–1202.
Murphy, B. R., Graham, B. S., Prince, G. A., Walsh, E. E., Channock, D. T., Karzon, D. T., Wright, P. F. Serum and nasal-wash immunoglobulin G and A antibody response of infants and children to respiratory syncytial virus F and G glycoproteins following primary infections. J. Clin. Microbiol. 23 (1986) 1009–1014.
Murphy, B. R., Alling, D. W., Snyder, M. H., Walsh, E. E., Prince, G. A., Channock, R. M., Hemming, V. G., Rodriguez, W. J., Kim, H. W., Graham, B. S., Wright, P. F. Effect of age and preexisting antibody on serum antibody response of infants and children to the F and G glycoproteins during respiratory syncytial virus infection. J. Clin. Microbiol. 24 (1986) 894–898.
Hall, C. B. Respiratory syncytial virus. In:Feign, R. D., Cherry, J. D. (eds.): Textbook of pediatric infectious diseases. W. B. Sounders, Philadelphia 1992, pp. 1633–1656.
Fulginiti, V. A., Eller, J. J., Sieber, O. F., Joyner, J. W., Minamitani, M., Meiklejohn, G. Respiratory virus immunisation: a field trial of two inactivated respiratory virus vaccines; an aqueous trivalent parainfluenza virus vaccine and an alum-precipitated respiratory syncytial virus vaccine. Am. J. Epidemiol. 89 (1969) 435–448.
Kapikian, A. Z., Mitchell, R. H., Chanock, R. M., Shvedoff, R. A., Stewart, C. E. An epidemiological study of altered clinical reactivity to respiratory syncytial (RS) virus infection in children previously vaccinated with an inactivated RS virus vaccine. Am. J. Epidemiol. 89 (1969) 405–421.
Kasel, J. A., Walsh, E. E., Frank, A. L., Baxter, B. D., Taber, L. H., Glezen, W. P. Relation of serum antibody to glycoproteins of respiratory syncytial virus with immunity to infection in children. Viral Immunol. 1 (1987/88) 199–205.
Toms, G. L., Webb, M. S. C., Milner, P. D., Milner, A. D., Routledge, E. G., Scott, R., Stokes, G. M., Swarbrick, A., Taylor, C. E. IgG and IgM antibodies to viral glycoproteins in respiratory syncytial virus infections of graded severity. Arch. Dis. Child. 64 (1989) 1661–1665.
Gimenez, H. B., Keir, H. M., Cash, P. Immunoblot analysis of human antibody response to respiratory syncytial virus infection. J. Gen. Virol. 68 (1987) 1267–1275.
Popow-Kraupp, T., Lakits, E., Kellner, G., Kunz, C. Immunoglobulin-class-specific immune response to respiratory syncytial virus structural proteins in infants, children, and adults. J. Med. Virol. 27 (1989) 215–223.
Allen, R. C., Dewez, P., Stuart, L., Gatenby, P. A., Sturgess, A. Antinuclear antibodies using HEp-2 cells in normal children and in children with common infections. J. Paediatr. Child Health 27 (1991) 39–42.
Cabral, D. A., Petty, R. E., Fung, M., Malleson, P. N. Persistent antinuclear antibodies in children without identifiable inflammatory rheumatic or autoimmune disease. Pediatrics 89 (1992) 441–444.
Tan, E. M., Chan, E. K. L., Sullivan, K. F., Rubin, R. L. Antinuclear antibodies (ANAs): diagnostically specific immune markers and clues toward the understanding of systemic autoimmunity. Clin. Immunol. Immunopathol. 47 (1988) 121–141.
Bürgin-Wolff, A., Signer, E., Friess, H. M., Berger, R., Birbaumer, A., Just, M. The diagnostic significance of antibodies to various cow's milk proteins (Fluorescent Immunosorbent test). Eur. J. Pediat. 133 (1980) 17–24.
Monce, N. M. Jr., Capel, V. L., Saqueton, C. B. A comparison of two fixatives on IFA HEp-2 slides for the detection of antinuclear antibodies. J. Immunoassay 15 (1994) 55–68.
Meddens, M. J. M., Herbrink, P., Lindeman, J., van Dijk, W. C. Serodiagnosis of respiratory syncytial virus (RSV) infection in children as measured by detection of RSV-specific immunoglobulins G, M, and A with enzyme-linked immunosorbent assay. J. Clin. Microbiol. 28 (1990) 152–155.
Ward, K. A., Lamden, P. R., Ogilvie, M. M., Watt, P. J. Antibodies to respiratory syncytial virus polypeptides and their significance in human infections. J. Gen. Virol. 64 (1983) 1867–1876.
Forster, J., Streckert, H. J., Werchau, H. The humoral immune response of children and infants to an RSV infection: its maturation and association with illness. Klin. Pädiatr. 207 (1995) 313–316.
Osiowy, C., Horne, D., Anderson, R. Antibody-dependent enhancement of respiratory syncytial virus infection by sera from young infants. Clin. Diagn. Lab. Immunol. 6 (1994) 670–677.
Giminez, H. B., Chisholm, S., Dornan, J., Cash, P. Neutralizing and enhancing activities of human respiratory syncytial virus-specific antibodies. Clin. Diagn. Lab. Immunol. 3 (1996) 280–286.
Conners, M., Kulkarni, A. B., Collins, P. L., Firestone, C., Holmes, K. L., Morse, H. C., Murphy, B. R. Resistance to respiratory syncytial virus (RSV) challenge induced by infection with a vaccinia virus recombinant expressing the RSV M2 protein (Vac-M2) is mediated by CD8+ T cells, while that induced by Vac-F or Vac-G recombinants is mediated by antibodies. J. Virol. 66 (1992) 1277–1281.
Chudwin, D. S., Ammann, A. J., Cowan, M. J., Wara, D. W. Significance of a positive antinuclear antibody test in a pediatric population. Am. J. Dis. Child 137 (1983) 1103–1106.
Schattner, A., Rager-Zisman, B. Virus-induced autoimmunity. Rev. Infect. Dis. 12 (1990) 204–222.
Meurman, O., Waris, M., Hedman, K. Immunoglobulin G antibody avidity in patients with respiratory syncytial virus infection. J. Clin. Microbiol. 30 (1992) 1479–1484.
Acrameas, S., Ternynck, T. The natural autoantibodies system: between hypotheses and facts. Mol. Immunol. 30 (1993) 1133–1142.
Berneman, A., Guilbert, B., Eschrich, S., Avrameas, S. IgG auto-and polyreactivities of normal human sera. Mol. Immunol. 30 (1993) 1499–1510.
Avrameas, S. Natural autoantibodies: from “horror autotoxicus” to “gnothi seauton”. Immunol. Today 12 (1991) 154–159.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Forster, J., Maier, O., Löbbert, J. et al. Prevalence of antibodies against HEp-2 cell antigen in infants and children hospitalized with respiratory syncytial virus infection. Infection 24, 407–411 (1996). https://doi.org/10.1007/BF01713039
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01713039