Abstract
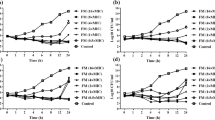
Two new fluoroquinolones (trovafloxacin and sparfloxacin) with enhanced activity against gram-positive pathogens and two earlier compounds (ciprofloxacin and ofloxacin) were tested for their in vitro inhibitory and bactericidal activity against 80 strains ofListeria monocytogenes. All strains were uniformly highly susceptible to trovafloxacin, the MIC90 being 0.25 mg/l. Resistance to sparfloxacin was not detected, however the MIC90 of sparfloxacin was eight times that of trovafloxacin. A few strains were resistant to ciprofloxacin and ofloxacin (MIC90 4 mg/l for both drugs). MBCs usually exceeded MICs by 2 to 4 times. The MBC90 of trovafloxacin (1 mg/l) was lower than that of the other three drugs (8 mg/l). After checking their ability to enter and grow within human enterocyte-like Caco-2 cells, four strains were used to study the intracellular activity and eradicating power of the four quinolones. Trovafloxacin was more active than sparfloxacin and the earlier fluoroquinolones in terms of both intracellular killing and inhibition of a cytopathogenic effect. The uniform high-level activity of trovafloxacin againstListeria monocytogenes isolates in conventional in vitro assays and its extracellular and intracellular killing of invasive strains suggest that this and maybe other new fluoroquinolones should be further investigated as possible anti-listerial agents.
Similar content being viewed by others
References
Schuchat A, Swaminathan B, Broome CV: Epidemiology of human listeriosis. Clinical Microbiology Reviews 1991, 4: 169–183.
Lorber B: Listeriosis. Clinical Infectious Diseases 1997, 24: 1–9.
Farber JM, Peterkin PI:Listeria monocytogenes, a foodborne pathogen. Microbiological Reviews 1991, 55: 476–511.
Schuchat A, Deaver KA, Wenger JD, Plikaytis BD, Mascola L, Pinner RW, Reingold AL, Broome CV, and the Listeria Study Group: Role of foods in sporadic listeriosis. I: Case-control study of dietary risk factors. Journal of the American Medical Association 1992, 267: 2041–2045.
Pinner RW, Schuchat A, Swaminathan B, Hayes PS, Deaver KA, Weaver RE, Plikaytis BD, Reeves M, Broome CV, Wenger JD, and the Listeria Study Group: Role of foods in sporadic listeriosis. II: Microbiologie and epidemiologic investigation. Journal of the American Medical Association 1992, 267: 2046–2050.
Jones EM, MacGowan AP: Antimicrobial chemotherapy of human infection due toListeria monocytogenes. European Journal of Clinical Microbiology & Infectious Diseases 1995, 14: 165–175.
Hof H, Nichterlein T, Kretschmar M: Management of listeriosis. Clinical Microbiology Reviews 1997, 10: 345–357.
Charpentier E, Gerbaud G, Jacquet C, Rocourt J, Courvalin P: Incidence of antibiotic resistance inListeria species. Journal of Infectious Diseases 1995, 172: 277–281.
Marget W, Seeliger HPR:Listeria monocytogenes: therapeutic possibilities and problems. Infection 1988, 16, Supplement 2: 175–177.
Southwick FS, Purich DL: Intracellular pathogenesis of listeriosis. New England Journal of Medicine 1996, 334: 770–776.
van den Broek PJ: Antimicrobial drugs, microorganisms, and phagocytes. Reviews of Infectious Diseases 1989, 11: 213–245.
Hof H: Intracellular microorganisms: a particular problem for therapy. Infection 1991, 19, Supplement 4: 193–194.
Piddock LJV: New quinolones and gram-positive bacteria. Antimicrobial Agents and Chemotherapy 1994, 38: 163–169.
Eliopoulos GM: In vitro activity of fluoroquinolones against gram-positive bacteria. Drugs 1995, 49, Supplement 2: 48–57.
Nakamura S, Minami A, Nakata K, Kurobe N, Kouno K, Sakaguchi Y, Kashimoto S, Yoshida H, Kojima T, Ohue T, Fujimoto K, Nakamura M, Hashimoto M, Shimizu M: In vitro and in vivo antibacterial activity of AT-4110, a new broad-spectrum quinolone. Antimicrobial Agents and Chemotherapy 1989, 33: 1167–1173.
Kojima T, Inoue M, Mitsuhashi S: In vitro activity of AT-4110 against clinical bacterial isolates. Antimicrobial Agents and Chemotherapy 1989, 33: 1980–1988.
Chin NX, Gu JW, Yu KW, Zhang YX, Neu HC: In vitro activity of sparfloxacin. Antimicrobial Agents and Chemotherapy 1991, 35: 567–571.
Briggs Gooding B, Jones RN: In vitro antimicrobial activity of CP-99,219, a novel azabicyclo-naphthyridone. Antimicrobial Agents and Chemotherapy 1993, 37: 2615–2622.
Eliopoulos GM, Klimm K, Eliopoulos CT, Ferraro MJ, Moellering RC: In vitro activity of CP-99,219, a new fluoroquinolone, against clinical isolates of gram-positive bacteria. Antimicrobial Agents and Chemotherapy 1993, 37: 366–370.
Neu HC, Chin NX: In vitro activity of the fluoroquinolone CP-99,219. Antimicrobial Agents and Chemotherapy 1994, 38: 2615–2622.
Pine L, Weaver RE, Carlone GM, Pienta PA, Rocourt J, Goebel W, Kathariou S, Bibb WF, Malcolm GB:Listeria monocytogenes ATCC 35152 and NCTC 7973 contain a nonhemolytic, nonvirulent variant. Journal of Clinical Microbiology 1987, 25: 2247–2251.
Facinelli B, Varaldo PE, Toni M, Casolari C, Fabio U: Ignorance about listeria. British Medical Journal 1989, 299: 738.
Facinelli B, Giovanetti E, Varaldo PE, Casolari C, Fabio U: Antibiotic resistance in foodborne listeria. Lancet 1991, 338: 1272.
Facinelli B, Giovanetti E, Casolari C, Varaldo PE: Interactions with lectins and agglutination profiles of clinical, food, and environmental isolates ofListeria. Journal of Clinical Microbiology 1994, 32: 2929–2935.
Roberts MC, Facinelli B, Giovanetti E, Varaldo PE: Transferable erythromycin resistance inListeria spp. isolated from food. Applied and Environmental Microbiology 1996, 62: 269–270.
Swaminathan B, Rocourt J, Bille J:Listeria. In: Murray PR, Baron EJ, Pfaller MA, Tenover FC, Yolken RH (ed): Manual of clinical microbiology. American Society for Microbiology, Washington, DC, 1995, p. 341–348.
National Committee for Clinical Laboratory Standards: Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved standard M7-A4. NCCLS, Villanova, PA, 1997.
Comité de l'Antibiogramme de la Société Française de Microbiologie: Statement 1996 CA-SFM. Zone sizes and MIC breakpoints for non-fastidious organisms. Clinical Microbiology and Infection 1996, 2, Supplement 1: 46–49.
Cormican GC, Jones RN: In-vitro activity of trovafloxacin (CP 99,129) tested by two methods against 150 vancomycin-resistant enterococcal isolates. Journal of Antimicrobial Chemotherapy 1996, 37: 847–849.
National Committee for Clinical Laboratory Standards: Methods for determining bactericidal activity of antimicrobial agents. Tentative guidelines M26-T. NCCLS, Villanova, PA, 1992.
Pascual A, Garcia I, Ballesta S, Perea EJ: Uptake and intracellular activity of trovafloxacin in human phagocytes and tissue-cultured epithelial cells. Antimicrobial Agents and Chemotherapy 1997, 41: 274–277.
Appleman MD, Cherubin CE, Heseltine PNR, Stratton CW: Susceptibility testing ofListeria monocytogenes: a reassessment of bactericidal activity as a predictor for clinical outcome. Diagnostic Microbiology and Infectious Disease 1991, 14: 311–317.
Nichterlein T, Kretschmar M, Budeanu C, Bauer J, Linss W, Hof H: Bay Y 318, a new quinolone derivative, rapidly eradicatesListeria monocytogenes from infected mice and L929 cells. Antimicrobial Agents and Chemotherapy 1994, 38: 1501–1506.
Michelet C, Avril JL, Cartier F, Berche P: Inhibition of intracellular growth ofListeria monocytogenes by antibiotics. Antimicrobial Agents and Chemotherapy 1994, 38: 438–446.
Michelet C, Avril JL, Arvieux C, Jacquelinet C, Cartier F: Comparative activities of new fluoroquinolones, alone and in combination with amoxicillin, trimethoprim-sulfamethoxazole, or rifampin, against intracellularListeria monocytogenes. Antimicrobial Agents and Chemotherapy 1997, 41: 60–65.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Facinelli, B., Magi, G., Prenna, M. et al. In vitro extracellular and intracellular activity of two newer and two earlier fluoroquinolones againstListeria monocytogenes . Eur. J. Clin. Microbiol. Infect. Dis. 16, 827–833 (1997). https://doi.org/10.1007/BF01700413
Issue Date:
DOI: https://doi.org/10.1007/BF01700413