Summary
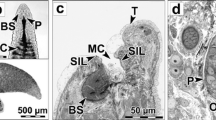
An azo dye technique was used to investigate localization of the acid hydrolase,β-glucuronidase, at light and electron microscope level in the stomach and digestive gland of the marine periwinkleLittorina littorea. Activity forβ-glucuronidase was located principally within digestive cells of the digestive gland and also associated with the microvillous border and epithelial cells lining the stomach. At the light microscope level all digestive tubules showed activity which appeared essentially restricted to the large heterolysosomes of the digestive cells. However not all digestive cells showed activity. In the electron microscope, reaction product was apparent in all types of macrovesicle in the digestive cells although not all stained positively. Heterophagosomes typically showed reaction product around their periphery or associated with the electron opaque contents. Activity was commonly seen around the apical edge of heterolysosomes where merging of heterophagosomes into heterolysosomes was apparent. Reaction product was commonly located within small electron lucent vesicles which lined the internal membrane of the heterolysosomes but sometimes also associated with flocculent, electron opaque contents. In the stomach dense clusters of reaction product were visible in lysosomes in the basal region of the epithelial cells and in the large granular inclusions of the secretory cells.
Similar content being viewed by others
References
BOWEN, I. D. (1971) A high resolution technique for the finestructural localization of acid hydrolases.J. Microsc. 94, 25–38.
FISHMAN, W. H., GOLDMAN, S. S. & DeLELLIS, R. (1967) Dual localization ofβ-glucuronidase in endoplasmic reticulum and in lysosomes.Nature, Lond. 213, 457–60.
FRETTER, V. & GRAHAM, A. (1962) British prosobranch molluscs: their functional anatomy and ecology. pp. 207–39. London: Ray Society.
FRETTER, V. & GRAHAM, A. (1976) A functional anatomy of invertebrates. pp. 472–549. London, New York, San Francisco: Academic Press.
MCQUISTON, R. W. (1969) Cyclic activity in the digestive diverticula ofLasaea rubra.Proc. malac. Soc. Lond. 38, 483–92.
MASON, A. Z. (1983) The uptake, accumulation and excretion of metals by the marine prosobranch gastropod molluscLittorina littorea (L.). Ph.D. Thesis, University of Wales.
MASON, A. Z. & NOTT, J. A. (1980) A rapid, routine technique for the X-ray microanalysis of microincinerated cryosections: an SEM study of inorganic deposits in tissues of the marine gastropodLittorina littorea (L.).J. Histochem. Cytochem. 28, 1301–11.
MASON, A. Z., SIMKISS, K. & RYAN, K. P. (1984) The ultrastructural localization of metals in specimens ofLittorina littorea collected from clean and polluted sites.J. mar. biol. Ass. UK 64, 699–720.
MERDSOY, B. & FARLEY, J. (1973) Phasic activity in the digestive gland cells of the marine prosobranch gastropod,Littorina littorea (L.).Proc. malac. Soc. Lond. 40, 473–82.
MOORE, M. N. & HALTON, D. W. (1977) The cytochemical localization of lysosomal hydrolases in the digestive cells of littorinids and changes induced by larval trematode infection.Z. Parasitenk. 53, 115–22.
MOORE, M. N., PIPE, R. K. & FARRAR, S. V. (1982) Lysosomal and microsomal responses to environmental factors inLittorina littorea from Sullom Voe.Mar. pollut. Bull. 13, 340–5.
MOORE, M. N., PIPE, R. K., FARRAR, S. V., THOMSON, S. & DONKIN, P. (1986) Lysosomal and microsomal responses to oil-derived hydrocarbons inLittorina littorea. InOceanic Processes in Marine Pollution — Biological Processes and Waste in the Ocean, Vol. 1 (edited by CAPUZZO, J. M. and KESTER, O. R.) Krieger: Melbourne (Florida), in press.
OWEN, G. (1970) The fine structure of the digestive tubules of the marine bivalveCardium edule.Phil. Trans. R. Soc. Ser. B 258, 245–60.
OWEN, G. (1972) Lysosomes, peroxisomes and bivalves.Sci. Prog. Oxf. 60, 299–318.
PAL, S. G. (1972) The fine structure of the digestive tubules ofMya arenaria (L.) II. Digestive cell.Proc. malac. Soc. Lond. 40, 161–70.
PIPE, R. K. & MOORE, M. N. (1985) The ultrastructural localization of lysosomal acid hydrolases in developing oocytes of the common marine mussel (Mytilus edulis).Histochem. J. 17, 939–49.
PIPE, R. K. & MOORE, M. N. (1986) An ultrastructural study on the effects of phenanthrene on lysosomal membranes and distribution of the lysosomal enzymeβ-glucuronidase in digestive cells of the periwinkleLittorina littorea. Aquatic Toxicology 8 (in press).
PLATT, A. M. (1971) Studies on the digestive diverticula ofMytilus edullis (L.). Ph.D. Thesis, Queen's University, Belfast.
SMITH, R. E. & FISHMAN, W. M. (1969)p-(Acetoxymercuric) aniline diazotate, a reagent for visualizing the naphthol AS—BI product of acid hydrolase action at the level of the light and electron microscope.J. Histochem. Cytochem. 17, 1–22.
STORRIE, B., POOL Jr, R. R., SACHDEVA, M., MAUREY, K. M. & OLIVER, C. (1984) Evidence for both prelysosomal and lysosomal intermediates in endocytic pathways.J. Cell Biol. 98, 108–15.
SUMNER, A. T. (1966) The cytology and histochemistry of the digestive gland cells of some freshwater lamellibranchs.J. R. microsc. Soc. 85, 201–11.
WERB, Z. & DINGLE, J. T. (1976) Lysosomes as modulators of cellular functions. Influence on the synthesis and secretion of non-lysosomal materials. InLysosomes in biology and pathology, Vol. 5 (edited by DINGLE, J. T. and DEAN, R. T.), pp. 127–56. North-Holland: Amsterdam, Oxford.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pipe, R.K. Light and electron microscope localization ofβ-glucuronidase activity in the stomach and digestive gland of the marine gastropodLittorina littorea . Histochem J 18, 175–183 (1986). https://doi.org/10.1007/BF01676118
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01676118