Abstract
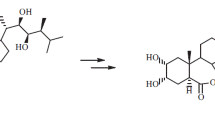
Synthesis of sterols with varying side chains, including deuterium labeled stigmasterol and sitosterol may be performed via the Wittig-Horner coupling of a 22 aldehyde derived from stigmasterol and a suitable sulfoxide derivative of the side chain. The X-ray structure determination of this synthon have been performed since it is a crucial step in order to know the absolute configuration of the chiral carbon atoms. Crystallographic data were as follows:a=7.437(2),b=10.103(4),c=10.274(3)Å, β=100.32(6)o, =759.4 Å3, space group P21 (No.4),Z=2,D c=1.239 g cm−3.
Similar content being viewed by others
References
Krajewski-Bertrand, M. A.; Milon, A.; Hartmann, M. A.Chem. Phys. Lipids 1992,63, 235.
Leonard, A.; Milon, A.; Krajewski-Bertrand, M. A.; Dufourc, E. J.Bull. Magn. Res. 1993,15, 124.
Schuler, I.; Duportail, G.; Glasser, N.; Benveniste, P.; Hartmann, M. A.Biochim. Biophys. Acta 1990,1028, 82.
Schuler, I.; Milon, A.; Nakatani, Y.; Ourisson, G.; Albrecht, A. M.; Benveniste, P.; Hartmann, M. A.Proc. Natl. Acad. Sci. USA. 1991,88, 6926.
Sheldrick, G. M.Crystallographic Computing 3; Oxford University Press: England,1985; pp 175–179.
Watkin, D.J.; Carruthers, J.R.; Betteridge, P.W.Crystals User Guide. Chemical Crystallography Laboratory; University of Oxford: England, 1985.
Johnson, C. K.ORTEP II, Oak Ridge National Laboratory: TN, 1976.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Marsan, M.P., Muller, I., Milon, A. et al. X-ray structure determination of a chiral synthon, essential for the synthesis of 25-2H-stigmasterol. J Chem Crystallogr 25, 783–786 (1995). https://doi.org/10.1007/BF01670336
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01670336