Abstract
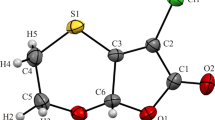
4-Amino-p-chlorobutyric acid lactam crystallizes as racemic crystals in the monoclinic space groupP21/n, witha=5.006(2),b=6.783(2),c=27.652(2) Å, β=92.25(1)°,Z=4,R=0.035 for 1618 unique reflections. As both enantiomers are present in a single crystal it is not possible to effect enantiomeric separation by crystal growth inhibition. This lactam may, however, be co-crystallized with (2R,3R)-(+)-tartaric acid forming a 2:1 complex in which only the (R) enantiomer is present. The complex crystallizes in the monoclinic space groupP21 witha=10.074(2),b=10.132(6),c=12.238(6) Å, β=99.13(2)°,Z=2,R=0.036 for 2291 unique reflections. Complex formation is effected by hydrogen bonding but enantioselectivity does not involve the chiral center of the title compound directly.
Similar content being viewed by others
References
Pasteur L.C.R. Acad. Sci. 1848,26, 535 Pasteur L.Ann. Chim. Res. 1850,28, 56.
Kandepud D.K.; Kaufman, R.J.; Singh, N.Science 1990,250, 975.
Mcbride J.M.; Carter R.L.Angew. Chem. Int. Ed. Engl. 1991,30, 293.
Inagaki, M.Chem. Pharm. Bull. 1977,25, 2497
Zaugg, H.E.J. Am. Chem. Soc. 1995,77, 2910.
Sato, N.; Uzuki, T.; Toi K; Agashi, T.Agric. Biol. Chem. 1969,33, 1107.
Brugidou, J.; Christol, H.; Sales, R.Bull. Soc. Chim. Fr. 1974, 2033.
Dolling, U.-H.; Douglas, A.W.; Grabowski, E.J.J.; Schoenewaldt, E.F.; Solar, P.; Sletzinger M.J. Org. Chem. 1978,43, 1634.
Velluz, L.; Armiand, G.; Joly, R.Bull. Soc. Chim. Fr. 1953, 342.
Ostromisslenskii, I.:Ber. Dtsch. Chem. Ges. 41, 3035.
Purvis, J.L.; US Patent 2 790 001, April 23 1957,Chem Abstr.,51, 1391 O1.
Addadi, L.; van Mil, J.; Lahav, M.J. Am. Chem. Soc. 1981,103, 1249.
Addadi, L.; Berkovitch-Yellin, Z.; Weissbuch, I.; Lahav, M.; Leiserowitz, L.Top. Stereochem. 16, I.
Addadi, L.; Weinstein, S.; Gati, E.; Weissbuch, I.; Lahav, M.J. Am. Chem. Soc. 1982,104, 4610.
Barton, D.H.; Kirby, G.W.J. Chem. Soc. 1962, 806.
Cope, A.C.; Ganellin, C.R.; Johnson, Jr. H.W.J. Am. chem. Soc. 1962,84, 3191.
Cope, A.C.; Ganellin, C.R.; Johnson Jr., H.W.; Auken, T.V.; Winkler, H.J.S.J. Am. Chem. Soc. 1963,85, 3276.
Hulshof, L.A.; Mckervey, M.A.; Wynberg, H.J. Am. Chem. Soc. 96, 3906.
Alcock, N.W.; Brown, J.M.; Hulmes, D.I.Tetrahedron: Asymmetry 1993,4, 743.
Schlenk Jr., W.Angew. Chem. Int. Ed. Engl. 4, 139.
Arad-Yellin, R.; Green, B.S.; Knossow, M.; Tsoucaris, G.Enantiomeric Selectivity of Host Lattices; Atwood, J.L.; Davies, J.E.D.; MacNicol, D.D.; Eds.; Academic Press: London, 1984; Inclusion compounds Vol. 3, Chap. 9.
Worsch, D.; Vögtle, F.Top. Curr. Chem. 1987,140, 21.
Toda, F.;Top. Curr. Chem. 1987,140, 43.Tsoucaris, G.Clathrates, Organic Solid State Chemistry; Desiraju, G.R., Ed.: Elsevier, Amsterdam, 1987; Chap. 7.
Wilen, S.H.Top. Stereochem. 1971,6, 107.
Sheldrick, G.M.SHELX-86, Crystallographic Computing 3; Sheldrick, G.M.; Kruger, C.; Goddard, R., Eds., Oxford University Press, London, 1985; p 175.
Sheldrick, G.M. SHELXL-93: A program for crystal structure determination.J. Appl. Cryst.,1993
Flack, H.D.; Schwarzenbach, D.Acta Crystallogr. 1988, A44, 499.
Weissbuch, I.; Popovitz-Biro, R.; Lahav, M.; Leiserowitz, L.Acta. Crystallogr. 1995, B51, 115–148.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Caira, M.R., Nassimbeni, L.R., Scott, J.L. et al. Resolution of optical isomers of 4-amino-p-chlorobutyric acid lactam by co-crystallization. J Chem Crystallogr 26, 117–122 (1996). https://doi.org/10.1007/BF01669727
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01669727