Abstract
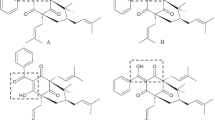
13α-Hydroxy-α-isolupanine (I) and 13β-hydroxy-α-isolupanine (II) crystallize in the space groupP212121 witha=13.123(2),b=13.221(3),c=8.224(1) Å,Z=4 anda=9.745(2),b=11.383(1),c=12.710(2) Å,Z=4, respectively. Structural differences between the epimeric molecules are concentrated around ringA. In (I) ringA has a half-chair conformation, and in (II) it is a distorted sofa. TheA/B ring junction configuration isquasi-trans in the case of (I) andquasi-cis in (II). The above differences can be explained by the different intermolecular interactions observed in these two structures.
Similar content being viewed by others
References
Bucourt, R. (1974)Topics in Stereochemistry, Vol. 8, Eliel, E. L., and Allinger, N. L., eds. (Interscience, New York).
Cruickshank, D. W. J., and Robertson, A. P. (1953)Acta Cryst. 6, 698.
Duax, W. L., and Norton, D. A. (1975)Atlas of Steroid Structure, Vol. 1 (Plenum Press, New York).
Duax, W. L., Weeks, C. M., and Rohrer, D. C. (1976)Stereochemistry, Vol. 9, Eliel, E. L., and Allinger, N. L., eds. (Wiley-Interscience, New York).
Kałuski, Z., Skolik, J., and Wiewiórowski, M. (1979) Proc. Pre-Congr. Symp. Org. Cryst. Chem. Poznań-Dymaczewo, July 30 to August 2, 1978.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pyżalska, D., Gawron, M. & Borowiak, T. Conformational analysis of 13-hydroxy-α-isolupanine epimers. Journal of Crystallographic and Spectroscopic Research 13, 31–41 (1983). https://doi.org/10.1007/BF01666794
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01666794