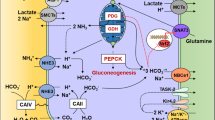
Summary
Whereas traditionally in acid-base physiology one considers just two organs (lungs and kidneys) to be involved in the regulation of systemic acid-base homeostasis, recent developments indicate that also the liver must be viewed as an important organ for pH regulation. This is because urea synthesis is a quantitatively important bicarbonate-consuming process, which in turn underlies a feedback control by the acid-base status at least in vitro. Consequently, renal ammoniagenesis, generally accepted to be a direct bicarbonate-generating process, can be viewed as a pH-controlled ammonium homeostatic response. In view of the controversies regarding the roles of ureogenesis and renal ammoniagenesis in acid-base regulation, their relationships were studied in 28 patients with normal renal functions, but varying degrees of a well-compensated chronic liver disease. Progressive loss of urea cycle capacity (as determined byin vitro incubations of human liver tissue) was parallelled by increasingin vivo plasma bicarbonate levels (and metabolic alkalosis) and an increasing NH +4 excretion into the urine. Accordingly, renal ammoniagenesis rose with the extent of metabolic alkalosis. Neither hypokalemia, hyperaldosteronism, diuretic treatment, or volume contraction were present, and a satisfactory explanation for this unusual behavior of renal ammoniagenesis in terms of traditional acid-base physiology cannot be given. Here, it seems that renal ammoniagenesis is governed rather by the need to eliminate ammonia than by the acid-base status. The data are compatible with a hepatic role in acid-base regulation: hyperbicarbonatemia results from a diminished hepatic HCO −3 disposal when urea synthesis is impaired and NH +4 homeostasis is maintained by the kidney via an increased NH +4 excretion into urine despite the presence of alkalosis.
Similar content being viewed by others
References
Ansley JD, Isaacs JW, Rikkers LF, Kutner MH, Nordlinger BM, Rudman D (1978) Quantitative tests for nitrogen metabolism in cirrhosis: relation to other manifestations of liver disease. Gastroenterology 75:570–579
Aruda JAL, Kurtzman NA (1977) Metabolic acidosis and alkalosis. Clin Nephrol 7:201–215
Atkinson DE, Camien MN (1982) The role of urea synthesis in the removal of metabolic bicarbonate and the regulation of blood pH. Curr Topics Cell Reg 21:261–302
Atkinson DE, Bourke E (1984) The role of ureagenesis in pH homeostasis. Trends Biochem Sci 9:297–300
Atkinson DE, Bourke E (1987) Metabolic aspects of the regulation of systemic pH. Am J Physiol 252:F947–956
Bean ES, Atkinson DE (1984) Regulation of the rate of urea synthesis in liver by extracellular pH. A major factor in pH homeostasis in mammals. J Biol Chem 259:1552–1559
Baertl JM, Sancetta SM, Gabuzda GJ (1963) Relation of acute potassium depletion to renal ammonium metabolism in patients with cirrhosis. J Clin Invest 42:696–706
Bergmeyer HU (ed) (1974) Methoden der Enzymatischen Analyse. Verlag Chemie, Weinheim
Burton K (1969) Determination of DNA with the diphenylamine reaction. Meth Enzymol 105:163–167
Casey TH, Summerskill WHJ, Bickford RG, Rosevear JW (1965) Body and serum potassium in liver disease. Relationships to arterial ammonia, blood pH and hepatic coma. Gastroenterology 48:208–215
Dölle W (1965) Der Säurebasenhaushalt bei Leberzirrhose. Hüthig, Heidelberg
Dustan HP, Corcoran AC, Page IH (1956) Renal function in primary aldosteronism. J Clin Invest 35:1357–1363
Gabuzda GJ, Hall PW (1966) Relation of potassium depletion to renal ammonium metabolism and hepatic coma. Medicine 45:481–490
Gerok W, Häussinger D (1984) Ammonia detoxication and glutamine metabolism in severe liver disease and its role in the pathogenesis of hepatic encephalopathy. In: Häussinger D, Sies H (eds) Glutamine metabolism in mammalian tissues. Springer, Heidelberg, pp 257–277
Gerok W, Häussinger D (1987) Neukonzeption der systemischen Säurebasenregulation — die Bedeutung der Leber. Internist 27:429–436
Haag G, Holldorf AW, Gerok W (1972) Veränderungen der Argininosuccinatsynthetase-Aktivität in der Leber bei chronischen Lebererkrankungen. Klin Wochenschr 50:887–889
Häussinger D (1983) Hepatocyte heterogeneity in glutamine and ammonia metabolism and the role of an intercellular glutamine cycle during ureogenesis in perfused rat liver. Eur J Biochem 133:269–274
Häussinger D, Gerok W (1985) Hepatic urea synthesis and pH regulation: role of CO2, HCO −3 , pH and the activity of carbonic anhydrase. Eur J Biochem 152:381–386
Häussinger D, Gerok W (1988) Urea synthesis and pH regulation. ISI Atlas Sci Biochem 1:65–70
Häussinger D, Gerok W, Sies H (1984) Hepatic role in pH regulation: role of the intercellular glutamine cycle. Trends Biochem Sci 9:300–302
Häussinger D, Gerok W, Sies H (1986) The effect of urea synthesis on extracellular pH in isolated perfused rat liver. Biochem J 236:261–265
Häussinger D, Meijer AJ, Gerok W, Sies H (1988) Hepatic nitrogen metabolism and acid-base homeostasis. In: Häussinger D (ed) pH Homeostasis. Academic, London, pp 337–377
Häussinger D (ed) (1988) pH Homeostasis. Academic Press, London
Halperin ML, Jungas RL (1983) Metabolic production and renal disposal of hydrogen ions. Kidney Int 24:709–713
Halperin ML, Goldstein MB, Stinebaugh BJ, Jungas RL (1985) Biochemistry and physiology of ammonium excretion. In: Seldin DW, Giebisch G (eds) The kidney: physiology and pathophysiology. Raven, New York, pp 1471–1490
Kaiser S, Gerok W, Häussinger D (1988) Ammonia and glutamine metabolism in human liver slices: new aspects on the pathogenesis of hyperammonemia in chronic liver disease. Eur J Clin Invest 18:535–542
Kathra BS, Smith RB, Milikan W, Sewell CW, Warren WD, Rudman D (1974) Activities of Krebs-Henseleit enzymes in normal and cirrhotic human liver. J Lab Clin Med 84:708–715
Knepper MA (1988) Renal transport of ammonium in systemic pH regulation. In: Häussinger (ed) pH homeostasis. Academic, London, pp 305–322
Krebs HA, Henseleit K (1932) Untersuchungen über die Harnstoffbildung im Tierkörper. Klin Wochenschr 11:757–759
Lemann J, Lennon EJ, Goodman AD, Litzow JR, Relman AS (1963) The net balance of acid in subjects given large loads of acid or alkali. J Clin Invest 44:507–517
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) The use of Folin's reagent for the determination of protein content in biological samples. J Biol Chem 193:265–275
Maier KP, Talke A, Gerok W (1979) Activities of urea cycle enzymes in chronic liver disease. Klin Wochenschr 57:661–664
Mullen KD, Denne SC, McCullough AJ, Savin SM, Bruno D, Tavill AS, Kalhan SC (1986) Leucine metabolism in stable cirrhosis. Hepatology 6:622–630
Oliver J, Bourke E (1975) Adaptations in urea and ammonium excretion in metabolic acidosis in the rat: a reinterpretation. Clin Sci Mol Med 48:515–520
Oster JR (1983) Acid-base homeostasis and liver disease. In: Epstein M (ed) The kidney in liver disease. Elsevier Science, New York, pp 147–182
Oster JR, Perez GO (1986) Acid-base disturbances in liver disease. J Hepatol 2:299–306
Pitts RF (1972) Control of renal production of ammonia. Kidney Int 1:297–305
Record CO, Iles RA, Cohen RD, Williams R (1975) Acidbase and metabolic disturbances in fulminant hepatic failure. Gut 16:144–149
Relman AS, Lennon EJ, Lemann J (1961) Endogenous production of fixed acid and the measurement of the net balance of acid in normal subjects. J Clin Invest 40:1621–1630
Rudman D, DiFulco JT, Galambos JT, Smith R, Slam A, Warren WD (1973) Maximal rates of excretion and synthesis of urea in normal and cirrhotic subjects. J Clin Invest 52:2241–2249
Rypins EB, Henderson JM (1980) A tracer method for measuring rate of urea synthesis in normal and cirrhotic subjects. Gastroenterology 78:1419–1424
Saad A, Gabuzda GJ (1964) Effect of potassium chloride supplements on thiazide induced hypokalemia alkalosis in patients with ascites. Gastroenterology 46:308
Schwab M, Damaschke H (1962) Atmung, Säure-Basen-Gleichgewicht und Ammoniak/Ammonium in Blut und liquor cerebrospinalis bei Leberzirrhose. Klin Wochenschr 40:184–199
Schwartz WB, Relman AS (1953) Metabolic and renal studies in chronic potassium depletion resulting from overuse of laxatives. J Clin Invest 32:258–271
Schwartz WB, Cohen JJ (1978) The nature of the renal response to chronic disorders of acid-base equilibrium. Am J Med 64:417–428
Tannen RL (1970) The effect of uncomplicated potassium depletion on urine acidification. J Clin Invest 49:813–827
Tannen RL (1977) Relationship of renal ammonia production and potassium homeostasis. Kidney Int 11:453–465
Tannen RL (1978) Ammonia metabolism. Am J Physiol 235:F265–277
Tannen RL, Sastrasinh S (1984) Response of ammonia metabolism to acute acidosis. Kidney Int 25:1–10
Ugarte G, Pino ME, Valenzuela J, Lorca F (1963) Urea cycle enzymatic abnormalities in patients with endogenous hepatic coma. Gastroenterol 45:182–188
Vilstrup H (1980) Synthesis of urea after stimulation with amino acids: relation to liver function. Gut 21:990–995
Walser M (1986) Roles of urea production, ammonium excretion and amino acid oxidation in acid-base balance. Am J Physiol 250:F181-F190
Welbourne T, Weber M, Bank N (1972) The effect of glutamine administration on urinary ammonium excretion in normal subjects and patients with renal disease. J Clin Invest 51:1852–1860
Author information
Authors and Affiliations
Additional information
This study was supported by Deutsche Forschungsgemeinschaft. Sonderforschungsbereich 154 „Experimentelle und Klinische Hepatologie“ and the Heisenberg Programm
Rights and permissions
About this article
Cite this article
Häussinger, D., Steeb, R. & Gerok, W. Ammonium and bicarbonate homeostasis in chronic liver disease. Klin Wochenschr 68, 175–182 (1990). https://doi.org/10.1007/BF01649081
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01649081